EXPERT INSIGHT
Chasing zero with COVID-19 vaccines
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has led to over 4 million deaths worldwide since late 2019.1,2 In an attempt to suppress the devastating pandemic, safe and effective vaccines are critical.3 According to the World Health Organization (WHO), since the first mass vaccination programme started in late 2020, as many as 13 different vaccines have been administered currently.4 In a recent interview with Omnihealth Practice, Professor Cowling, Benjamin John shared his insights over the role of vaccination under the local pandemic control policy.
The role of immunogenicity in COVID-19 vaccination
The effectiveness of COVID-19 vaccine depends on both antibody-mediated and T-cell-mediated immunity.5 In principle, neutralizing antibodies block the virus from entering the host cells to limit the infection, playing a protective role at the later stage of infection, and preventing the relapse of infection.6 In contrast, T-cell immune responses are observed inside infected cells, with a vital role in clearing and cleaning the viral-infected cells.6 Therefore, it is suggested that while the magnitude of neutralizing antibody responses relates to the prevention of infection, T-cell immunity is associated with protection against severe disease from COVID-19. However, cautions should be taken when evaluating the T-cell immunology of vaccines based on the available data, as the measuring and interpreting approaches for T-cell responses are not as standardized as for the antibody levels.
Safety and effectiveness of the locally available COVID-19 vaccines
There are two types of vaccines deployed in Hong Kong, namely the inactivated whole-virus vaccine and the BNT162b2 vaccine (lipid nanoparticle [LNP]-encapsulated messenger ribonucleic acid [mRNA] vaccine).7 In terms of safety, it is observed that mild and immediate reactions after vaccination, including sore arms, fever and tiredness, are more common for mRNA vaccines including the BNT162b2, especially after the second dose. Although data are not sufficient to estimate the risk of rare serious reactions for both vaccines currently, it is believed to be comparably low. As the benefits of vaccination outweigh the risk of side effects, high vaccine uptake is currently encouraged under the local public health policy.
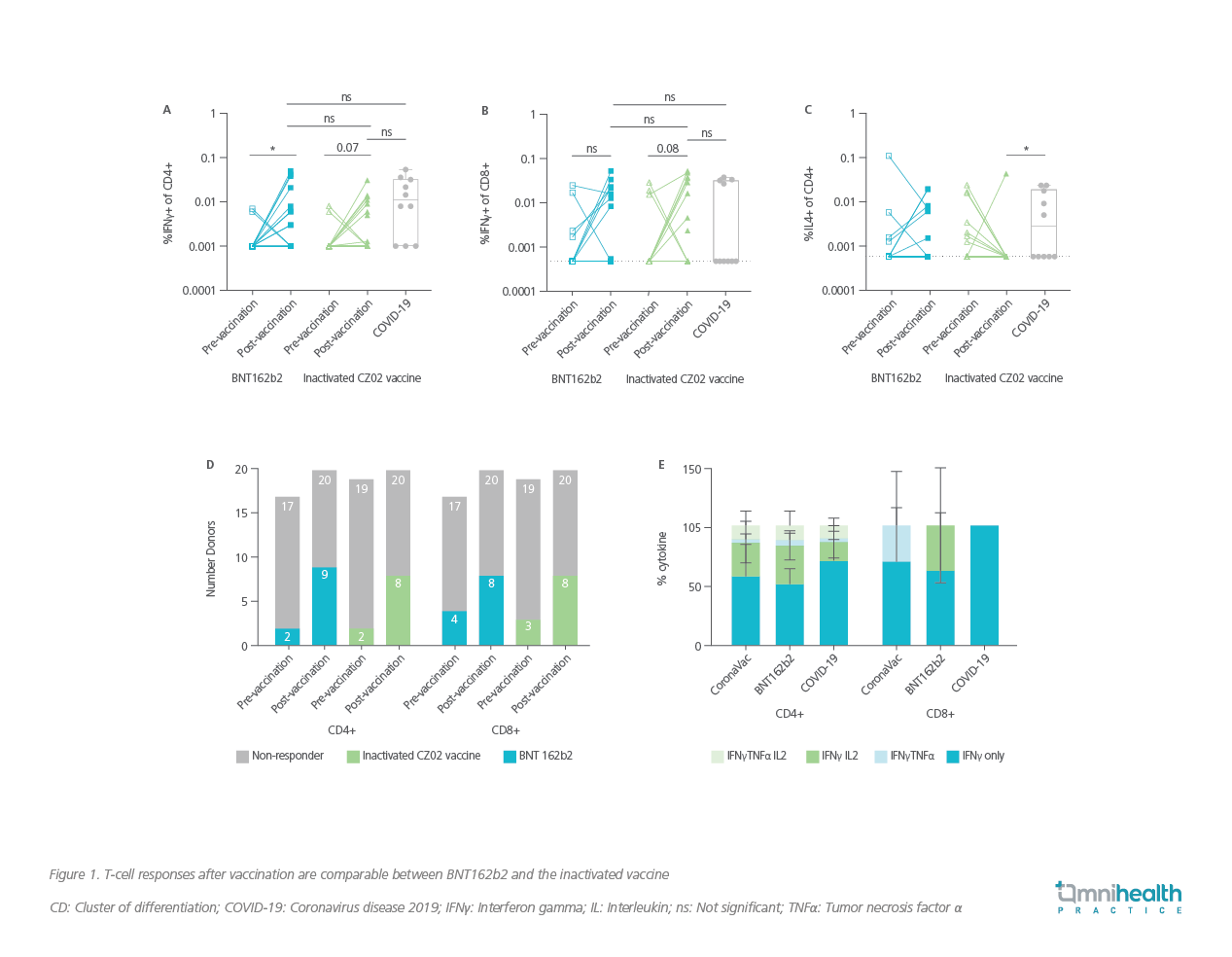
In terms of vaccine effectiveness, both vaccines induce similar protection against SARS-CoV-2 through T-cell immunity.8 In a comparative data on the immunogenicity of BNT162b2 vaccine (n=20) and the inactivated vaccine (n=20), both vaccines induced comparable cluster of differentiation (CD) 4 and CD8 T-cell responses to spike proteins at 1 month post 2-dose vaccination (Figure 1A, 1B, 1C), proportions of subjects that had detectable post-vaccination T-cell responses (Figure 1D), and post-vaccination production levels of polyfunctional cytokines by T-cells in vaccine responders (Figure 1E).8 Since T-cell responses are likely to be crucial to controlling the severe and fatal outcomes, the two vaccines are proposed to be both effective in preventing such adverse outcomes of SARS-CoV-2 infection.8
However, the two vaccines seem to induce different levels of prevention against viral infection as different concentrations of neutralizing antibodies were observed in the study.8 Results found that higher levels of SARS-CoV-2 specific binding and neutralizing antibody responses [in plaque reduction neutralization test (PRNT)50, PRNT90, surrogate virus neutralization (sVNT), spike receptor binding (RBD), spike N terminal domain (NTD) binding, and spike S2 domain binding antibody levels] were observed with the BNT162b2 vaccine (n=49) compared with the inactivated vaccine (n=49).8
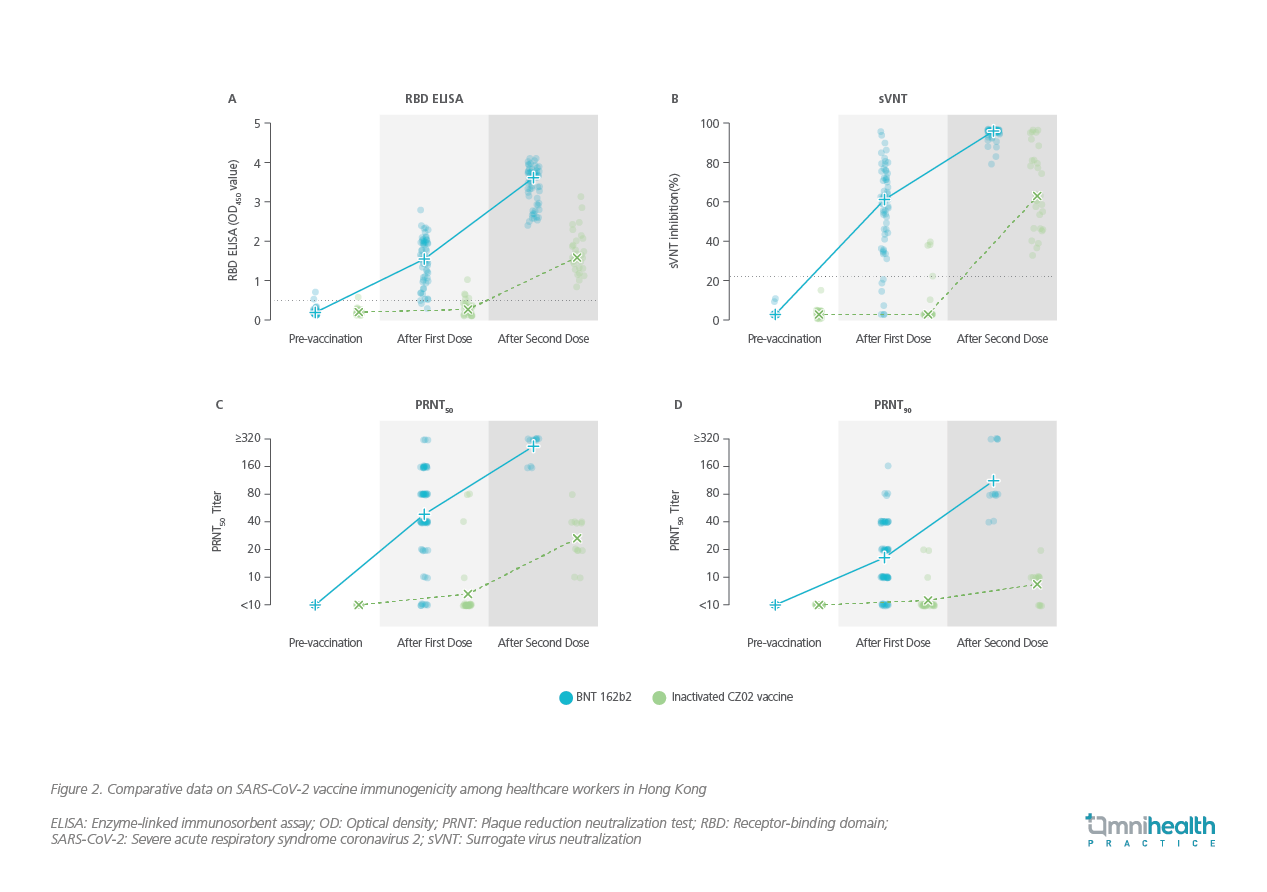
Similar results were also demonstrated in a recently published study involving a cohort of 1,442 local healthcare workers, where antibodies to SARS-CoV-2 were measured using enzyme-linked immunosorbent assay (ELISA), sVNT assay, and then a PRNT with live SARS-CoV-2 virus.9 The results suggested that antibody concentrations measured by ELISA and sVNT rose substantially after the first dose and then rose again after the second dose of the BNT162b2 vaccination (Figure 2A and 2B).9 In contrast, the recipients of the inactivated vaccine had low antibody concentrations by ELISA and sVNT after the first dose, which rose to the moderate levels after the second dose.9
Of note, in a subset of 12 participants who got PRNT results, after the second dose of vaccination, the geometric mean PRNT50 titre was 269 in the BNT162b2 group versus 27 in the inactivated vaccine group (Figure 2C); and the geometric mean PRNT90 titre was 113 in the BNT162b2 group versus 8.4 in the inactivated vaccine group (Figure 2D).9 Given that the difference in concentrations of neutralizing antibodies could translate into difference in protective effectiveness of the vaccines, the results might indicate a need for future studies exploring alternative strategies, including administration of booster doses, to increase antibody concentrations and clinical protection in recipients of the inactivated vaccines.9
Further studies needed for COVID-19 booster doses
Further studies in the performance of booster doses, particularly the feasibility of mixing vaccine types in booster vaccination, are suggested from the previous efficacy data on the 2-dose regimen. It is suspected that mixing vaccine types may improve the overall protection against SARS-CoV-2 infection. For instance, when the immune system is exposed to a broader range of spike proteins with the inactivated vaccine in the initial 2 doses, the overall protective efficacy will be improved with subsequent mRNA boosting dose targeting the specific spike proteins.
The current local policy focuses on the initial doses of vaccination only. While data are barely available, especially with regard to the inactivated vaccines, for administrating booster doses with platforms different from the initial doses, trials should be conducted to evaluate the performance of boosters, as well as combining various types of vaccines in booster and initial doses, before implementing the booster vaccination policy. The government should also justify the decision clearly, preparing detailed timeline for regular administration of booster doses if the booster policy is to be implemented.
Insights over the current local vaccination policies
Currently, as of August 29, 2021, only 60.8% of the total eligible population in Hong Kong have received the first dose of COVID-19 vaccines and 50% with the second dose.10 A higher level of vaccination coverage is essential to lifting the public health measures, including travel restriction, gathering limits, social distancing, and the mandatory mask-wearing requirement. Indeed, these restrictions had been well-implemented in Hong Kong before starting the vaccination programs for better infectious control. Even though the vaccination programs are now in place, these measures are expected to continue and become a new normal.
Notably, amid Hong Kong’s battle against the fifth wave of COVID-19, the elderly aged 80 or above have a very low vaccination coverage (only 11.38% of individuals in this age group are vaccinated with first dose10), while they are more vulnerable to COVID-19 infection, comparing with other age groups. Personal perception towards the benefits and risks may account for the low vaccination rates. In view of the improving local pandemic situation and the media coverage of the serious side effects of the vaccines, the benefits of vaccination may be undermined locally when compared with other countries. That may explain why some people in Hong Kong, especially those healthy and younger population, are reluctant to get vaccinated.
Fortunately, the vaccination uptake improves along with the availability of more financial incentives, which include the apartment lottery and other giveaways. The mandatory regular COVID-19 testing for certain unvaccinated groups also helps boost the vaccination rates. A clear timeline for relaxing social distancing, hospital visiting, travel, and the mask-wearing policy, along with more incentives, including the shortening of quarantine, the vaccination coverage is expected to be further increased. Besides, addressing the concerns of side effects by providing more safety data from reliable sources, as well as inviting celebrities or charisma to promote the vaccine programs may also help.
However, vaccination boosting policies may not be the focus now, as the “zero-COVID” approach seems to be the priority of the local government, which focuses on controlling imported cases and other strategies to limit viral transmission in the local community. If the government wishes to re-open the borders with Mainland China, at least 70% vaccine coverage is required (as stated by the Mainland government).
Future disease control strategies in the local community
Similar to influenza, it is not possible to maintain zero cases of COVID-19 after relaxing the infection control measures, even with an adequate level of vaccination uptake in the local community. If “zero-COVID” approach still prevails in the coming year, travel restriction should be maintained with relaxed local control measures when there are no local cases. However, when countering the fifth wave of COVID-19, all these measures should be maintained. While maintaining “zero-COVID” may not be possible in the long term, the local community is expected to live alongside with the virus in the future. As such, the disease control measures shall be relaxed when a higher level of vaccination coverage is reached.
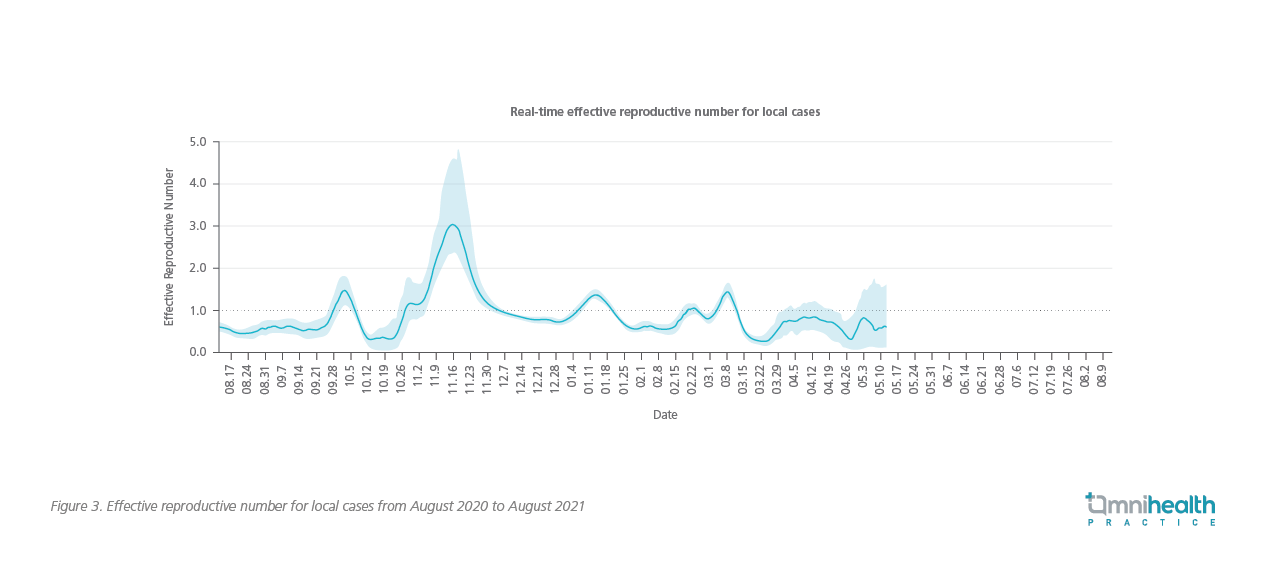
Besides, potential challenges still exist in controlling future disease transmission. Regarding the overwhelming situation of infection with the Delta variant of SARS-CoV-2 (B.1.617.2),11 it is believed that the current measures may not be sufficient to control the transmission. It is because when the fourth wave of COVID-19 outbreak (which arrived in late November 202012) nearly subsided in December 2020, the reproductive number for local cases reached only to a satisfactory level at 0.9 (Figure 3),13 suggesting that the same measures may not be sufficient when countering the fifth wave of virus, particularly under the threat of more contagious Delta variant. It is hoped that a higher vaccination coverage could help alleviate the spread of disease in the coming waves of virus, while stricter measures may still be required.
Message to physicians
The benefits of COVID-19 vaccines certainly far outweigh the risks. As it takes at least 5 to 6 weeks for full protection from vaccine, vaccination uptake should not be delayed, particularly in the face of the coming fifth wave of virus. Hong Kong has been successful in controlling the spread of COVID-19 in the past years, with probably the lowest prevalence in the world. If we fail to fully prepare for the next wave of virus and the Delta variant, the number of local cases is expected to be surged in a short period of time.
Conclusion
COVID-19 remains a major health burden in Hong Kong that requires disease control measures and extensive vaccination coverage. Through clinical trials supporting the effectiveness and safety of the vaccines, along with the policies and incentives that promote the vaccination, it is believed that the vaccination coverage in the public will be raised, and that the spread of disease will also be minimized.

