Research Spotlight
Breaking the myths: Consensus statements from local rheumatologists recommend covid-19 vaccination in patients with autoimmune rheumatic diseases
In brief
The Hong Kong Society of Rheumatology (HKSR) published a set of consensus statements for Coronavirus Disease 2019 (COVID-19) vaccination in adult patients with autoimmune rheumatic diseases (ARDs). This statement serves as a guide to local rheumatologists and other specialists, nurses, healthcare professionals and the public regarding the use of COVID-19 vaccines in patients with ARDs.
Background
ARD is a group of chronic medical illness characterized by systemic inflammation, in which a dysregulated immune system causes damage or dysfunction to connective tissues and leads to various treatment-related complications and comorbidities.1,2 In the current COVID-19 pandemic, patients with pre-existing chronic health conditions are more prone to serious illness and mortality from the infection, arousing concerns among patients with ARDs regarding their suitability for the COVID-19 vaccination.1,3
The concerns about the COVID-19 vaccines from the patient community were observed in a web-based questionnaire study, in which 32.2% of 1,266 patients with systemic rheumatic diseases reported uncertainty about the COVID-19 vaccination, and 13.6% of the population were unwilling to get vaccinated.4 The main concerns from the patients were the scarcity of experience and background information regarding the new COVID-19 vaccines, the use of a new vaccination technology, possible induction of a disease flare, and the risk of developing local reactions or side effects.4 However, the willingness to get vaccinated increased when the vaccination was recommended by specialists, with a reported uncertainty declined to 28.4% and unwillingness to 8.8%.4 This emphasizes the timely and critical involvement of rheumatologists in vaccination uptake, highlighting the importance of professional advice and accurate information education when encouraging COVID-19 vaccination.1,4
Considering the need of a guide to address patients’ concerns, the HKSR developed a comprehensive set of consensus statements for the use of COVID-19 vaccines among local patients with ARDs, clarifying the relationship between the pandemic, vaccination, ARDs, and immunosuppressive treatments.1
Methodology
Clinical questions regarding the COVID-19 vaccination in adult patients with ARDs were discussed by 27 core members of the HKSR, including council members and rheumatologist-in-charge of most public hospitals.1 The discussion was summarized into six clinical questions afterward, using the PICO (problem/population, intervention, comparison and outcomes) approach.1 A systemic literature search (SLR) in PubMed was subsequently conducted to identify the evidence for the consensus statements, covering only clinical trials, cohort studies and case series published in English from December 2019 to March 28, 2021.1
Prior to the Delphi exercise, the strength of evidence (SoR) and quality of evidence (QoE) were evaluated for each of the eight finalized statements based on the Grades of Recommendations Assessment, Development and Evaluation (GRADE) system (Table 1).1 Also provided with the results of the SLR and explanatory notes, full members of the HKSR were invited to anonymously indicate the level of agreement by arbitrary scores, provide feedback, indicate reasons for disagreement and suggest modifications for the statements.1 Statements were subjected to amendments and subsequent rounds of voting until at least 80% of Delphi participants agreed for all statements.1

Results and discussion
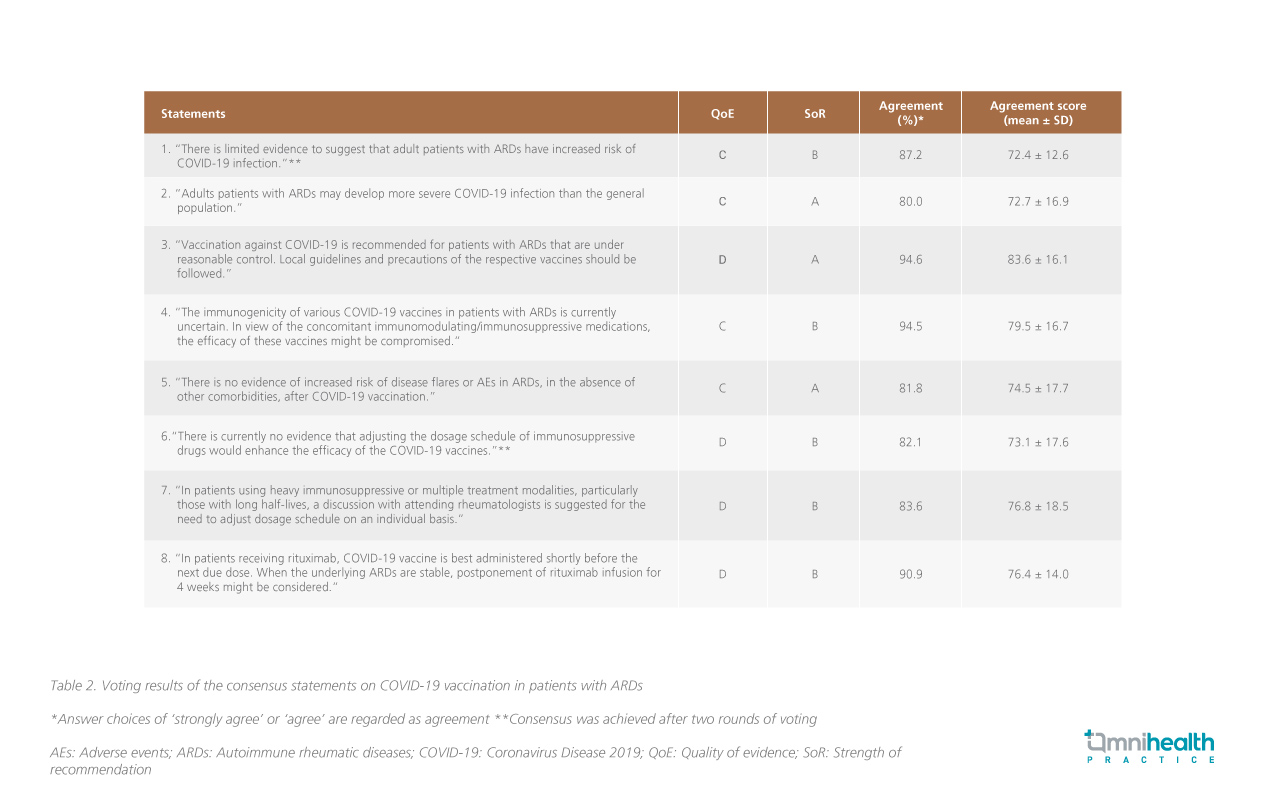
A total of 55 full members of HKSR participated in the Delphi exercise, representing more than 60% of the local practicing rheumatologists with the majority working in public hospitals.1 With consideration of the existing evidence and local perspectives such as the availability of specific vaccines and the outbreak situation, consensus of the statements was reached after two rounds of voting (Table 2).
Significant interpretation
Dr. So, Ho and Dr. Yip, Man-Lung Ronald, authors of the consensus statements as well as core members of the HKSR, noted that the consensus statements would address the concerns regarding the COVID-19 vaccines and encourage vaccination among patients with ARDs. Although there is growing evidence suggesting that immunosuppressive treatments would affect the antibody response to the COVID-19 vaccines, Dr. So noted that an antibody response may not reflect the efficacy of vaccines. Clinically, there is no evidence of COVID-19 vaccination increasing the ARD activity or the risk of acquiring vaccine-related side effects, such as facial paralysis, in adult patients with ARDs. “As the COVID-19 vaccines available in Hong Kong are not live-attenuated vaccines, the risk of developing COVID-19 infection through vaccination is not possible in patients with ARDs,” reiterated Dr. So when considering the safety of COVID-19 vaccination in patients with ARDs.
When mentioning the balance between the treatment for ARDs and COVID-19, Dr. Yip emphasized the importance of early treatment and the treat-to-target approach in ARD management, highlighting that adjustments in the ARDs treatment regimen in view of the COVID-19 pandemic, as well as the COVID-19 vaccination, are not recommended. “There is growing evidence showing that with earlier treatment, the disease control will be easier, leading to a better prognosis, higher remission rate, and lower occurrence of comorbidities and physical disabilities,” stated Dr. Yip. Dr. So also supported this opinion with guidelines from the United States, the European Union and Asia, stating that treatments for ARDs should not be discontinued in the pandemic as active rheumatic diseases also increase the risk of COVID-19 infection and severity. Importantly, ARD treatments involving immunosuppression should not be compromised for the vaccination. That said, postponement of rituximab infusion or COVID-19 vaccination may be considered as B-cell depleting therapy may reduce the protective immunity against severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) following COVID-19 vaccination.5 Patients with uncontrolled disease activity are also recommended to hold COVID-19 vaccination until their diseases are stable, Dr. Yip noted.
Both Dr. So and Dr. Yip expressed their wishes for the consensus statements to improve patients’ confidence towards COVID-19 vaccines. By participating in interviews, translating the statements into lay terms in Chinese language, and proactively addressing the concerns of the vaccines through patient support groups, they hoped that the local patient community will understand that the benefits of COVID-19 vaccination can actually outweigh the risks. Dr. So concluded, although more data regarding the efficacy of the vaccines is needed, it should not affect the decision on COVID-19 vaccination and the treatment for ARDs. Dr. Yip agreed, summarizing that “The discontinuation of routine treatment is not required to accommodate COVID-19 vaccination in most patients with ARDs. Also, as vaccination is generally beneficial to our patients during the current pandemic situation, we encourage patients with stable, controlled ARDs to receive COVID-19 vaccination.”

