MEETING HIGHLIGHT
First glimpse of zolbetuximab-based chemo-immunotherapy shows promise in CLDN18.2- and PD-L1-positive advanced G/GEJ cancer: ILUSTRO phase 2 data from ASCO GI 2026
Advanced gastric and gastroesophageal junction (G/GEJ) adenocarcinoma remains associated with a poor prognosis. With first-line (1L) chemotherapy, median overall survival (OS) is approximately 8.8-12.2 months, with many patients unable to receive second- or later-line therapies due to rapid disease progression and declining performance status.1,2 In this context, optimizing 1L treatment has become increasingly critical, particularly as molecular characterization has enabled more precise, biomarker-driven approaches.1,2 At the 2026 ASCO Gastrointestinal Cancers Symposium, Dr. Kohei Shitara presented updated efficacy and safety data from the phase 2 ILUSTRO trial, focusing on a triplet regimen combining zolbetuximab, chemotherapy, and PD-1 inhibition in patients with claudin-18.2 (CLDN18.2)-positive advanced GE/GEJ.3 Dr. Daniel Lin from the Thomas Jefferson University Hospital, United States further provided a broader perspective of the evolving role of CLDN18.2 as a clinically actionable biomarker in the frontline setting and highlighted the potential of biomarker-enriched combination strategies.2
Getting 1L therapy right: The role of CLDN18.2 in advanced gastric/GEJ cancer
For patients with advanced G/GEJ cancer, the choice of 1L therapy is critical, as it profoundly impacts subsequent treatment opportunities and overall survival.1,2 Real‑world evidence and clinical trial data consistently show substantial attrition across treatment lines, with only a minority of patients able to receive second‑ or later‑line therapies.1,2 This underscores the importance of maximizing therapeutic benefit in the 1L setting. The 1L treatment landscape of G/GEJ cancer becomes increasingly biomarker‑driven, CLDN18.2 has emerged alongside HER2 and PD‑L1 as an important validated therapeutic target.2 CLDN18.2 is a tight junction protein normally confined to the gastric mucosa but aberrantly expressed on the surface of malignant gastric and GEJ tumor cells.4,5
Evidence supporting CLDN18.2 as a therapeutic biomarker was first demonstrated in the phase 2 FAST study, where patients whose tumors expressed CLDN18.2 in ≥70% of cells with moderate to strong membranous staining by IHC experienced enhanced survival benefit. This cutoff was later standardized to
Co-expression of CLDN18.2 and PD-L1 is reported in 26%-79%, depending on assay and scoring thresholds.2 However, the optimal first-line strategy for dual-positive patients remains unclear.8 Guideline recommendations reflect this uncertainty: National Comprehensive Cancer Network (NCCN) now lists zolbetuximab + fluoropyrimidine/oxaliplatin as a Category 1 preferred 1L option for CLDN18.2-positive, HER2-negative tumors, while European Society for Medical Oncology (ESMO) and Chinese Society of Clinical Oncology (CSCO) recognize CLDN18.2-targeted therapy within biomarker-driven frameworks, but provide no clear guidance for integrating PD-1/PD-L1 inhibitors.9-11
Zolbetuximab, the first-in-class monoclonal antibody directed targeting CLDN18.2, exerts antitumor activity through antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity.2,4-6 Preclinical findings also suggest that CLDN18.2 blockade may modulate the tumor immune microenvironment—enhancing CD8⁺ T‑cell infiltration and improving antitumor activity when combined with PD‑1 inhibition.2 These provide a strong rationale for ILUSTRO, the first prospective trial designed to evaluate the combination of zolbetuximab with chemotherapy and PD-1 blockade in 1L dual-positive patients, providing the first clinical insights in this population.3
The ILUSTRO phase 2 trial: Evaluating zolbetuximab triplet therapy in the frontline
The multicohort phase 2 ILUSTRO trial was designed to evaluate zolbetuximab as monotherapy or in combination with chemotherapy and/or PD-1 inhibition in patients with CLDN18.2-positive advanced G/GEJ adenocarcinoma.3,5 The trial includes several cohorts exploring different treatment strategies, with cohort 4 specifically assessing zolbetuximab in combination with mFOLFOX6 + nivolumab in the 1L setting.2,3,5 According to Dr. Shitara, this represents the first dataset evaluating a triplet regimen of zolbetuximab, PD-1 inhibition, and chemotherapy specifically in dual-positive patients (CLDN18.2-positive and PD-L1 combined positive score [CPS]
Cohort 4 enrolled patients with previously untreated, HER2-negative, locally advanced unresectable or metastatic G/GEJ adenocarcinoma whose tumors demonstrated CLDN18.2 positivity by central immunohistochemistry.2,3 Other key eligibility criteria also included an Eastern Cooperative Oncology Group (ECOG) performance status of 0-1.2,3 Cohort 4 comprised a safety lead-in phase (cohort 4A) followed by a main efficacy cohort (cohort 4B).2,3 Cohort 4A evaluated the tolerability of zolbetuximab dosing in combination with nivolumab + mFOLFOX6, while cohort 4B was designed to further assess efficacy and safety, with a planned enrichment of patients with high CLDN18.2 expression (defined as
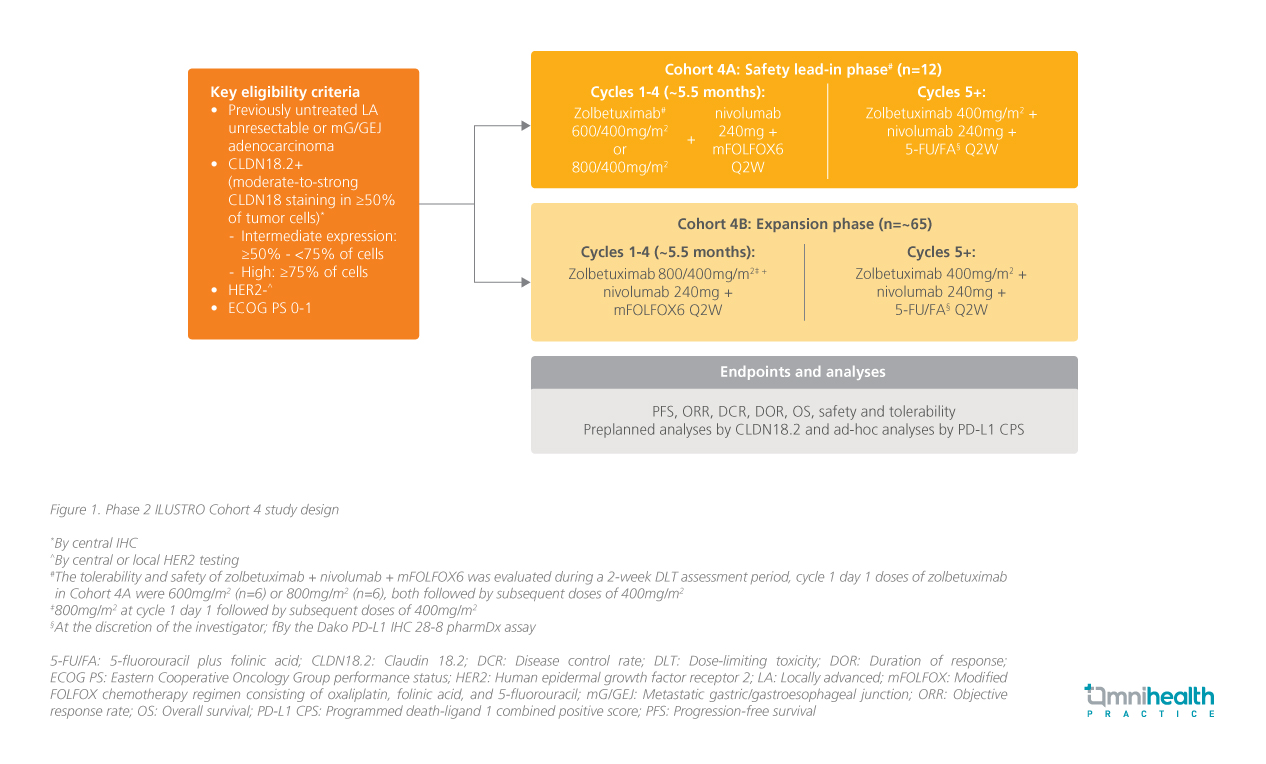
Encouraging efficacy signals in cohort 4B
Efficacy analyses from cohort 4B focused primarily on progression-free survival (PFS), with additional evaluation of objective response rate (ORR), duration of response, and OS.3 At the time of data cutoff, the median follow-up was 11.5 months (95% CI: 10.9-15.6), with PFS assessed by investigators per RECIST v1.1.3 A total of 12 patients were enrolled in cohort 4A and 71 patients in cohort 4B.3 Across both cohorts (n=77), the median age was 61.0 years (range: 37.0-86.0), 62.3% were male, and 79.1% were of Asian ethnicity.3 Most patients had PD-L1-positive disease (CPS
In cohort 4B, treatment with zolbetuximab + mFOLFOX6 + nivolumab resulted in a median PFS of 14.8 months (95% CI: 8.3-non estimable [NE]).3 The 6-month and 12-month PFS rates were 72.6% and 59.1%, respectively.3 Stratification by CLDN18.2 expression revealed a more pronounced benefit in patients with high CLDN18.2 expression.3 In this subgroup, median PFS was 18.0 months (95% CI: 11.1-NE), compared with 6.7 months (95% CI: 3.0-NE) in patients with intermediate expression.3 Further biomarker enrichment was observed when PD-L1 status was considered.3 Among patients with high CLDN18.2 expression and a PD-L1 CPS
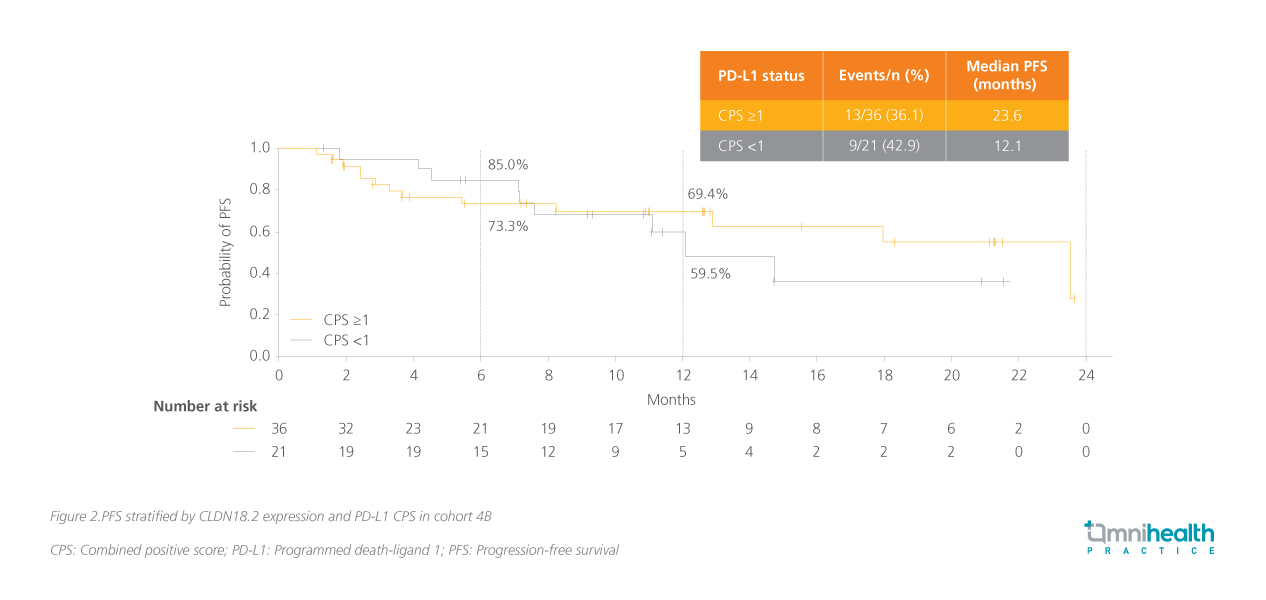
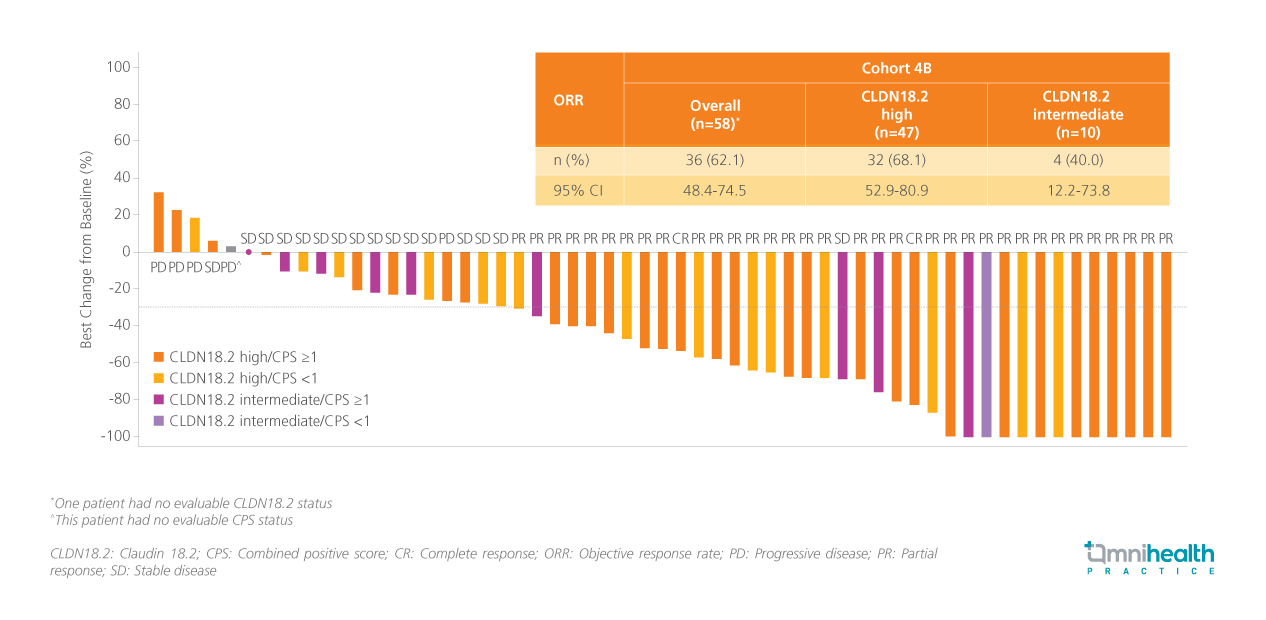
Tumor responses further supported the observed PFS benefit.3 In patients with measurable disease in cohort 4B, ORR was 62.1% (95% CI: 48.4-74.5) overall and 68.1% (95% CI: 52.9-80.9) in the high CLDN18.2 subgroup.3 Waterfall plot analyses demonstrated tumor shrinkage in a substantial proportion of patients, with many achieving at least a partial response (figure 3).3 Duration-of-response analyses indicated that many responses were durable beyond initial assessment, with a median of 19.1 months (95% CI: 10.8-NE), aligning with the favorable PFS outcomes.3 While OS data remained immature at the time of analysis, early trends appeared favorable in patients with high CLDN18.2 expression.3 However, no median OS had yet been reached, and longer follow-up is required before drawing conclusions on the survival benefit.3
Safety, tolerability, and the path forward for zolbetuximab-based combinations
In addition to efficacy, ILUSTRO cohort 4 provided important insights into the safety and tolerability of zolbetuximab triplet therapy in the frontline setting.3 Safety analyses included patients from both cohort 4A and cohort 4B who received zolbetuximab at a dose of 800mg/m2 on cycle 1 day 1 followed by 400mg/m2 in subsequent cycles, in combination with nivolumab + mFOLFOX6.3 The median duration of treatment was 288.5 days (range: 1-1,271) for zolbetuximab and 226.0 days (range: 1-872) for nivolumab.3 Mean relative dose intensity was high for both agents, at 97.1% for zolbetuximab and 97.0% for nivolumab, reflecting that patients were able to receive nearly all planned doses.3
The overall safety profile of the triplet regimen was manageable, with no unexpected safety signals observed.3 Treatment-emergent adverse events (TEAEs) led to discontinuation of any study drug in 49.4% of patients, while discontinuation of zolbetuximab occurred in 5.2%.3 TEAEs were consistent with the known safety profiles of zolbetuximab or nivolumab + mFOLFOX6.3 Nausea (80.5%) and vomiting (37.7%), common concerns with zolbetuximab, were mostly low grade, with Grade 3 events reported in 0% and 3.9%, respectively.3,5,12 Other frequently observed TEAEs included decreased appetite (72.7%) and decreased neutrophil count (45.5%).3 “From a tolerability standpoint, the triplet regimen was feasible, with safety findings consistent with prior experience,” Dr. Shitara commented.
Guideline alignment and phase 3 clinical trial pipeline
Taken together, the efficacy and safety findings from ILUSTRO cohort 4B provide a strong rationale for further investigation in a larger, randomized setting.2,3 These phase 2 data have directly informed the design of the ongoing phase 3 LUCERNA trial (NCT06901531), which is evaluating zolbetuximab in combination with pembrolizumab and chemotherapy as 1L treatment for patients with CLDN18.2-positive, HER2-negative, PD-L1-positive locally advanced unresectable or metastatic G/GEJ adenocarcinoma.2,3 The LUCERNA study will assess whether the promising signals observed in ILUSTRO translate into meaningful clinical benefit in a larger, globally representative population.2,3
This biomarker-enriched trial approach reflects the broader shift toward precision medicine in advanced G/GEJ cancers, consistent with current ESMO and NCCN guidance emphasizing comprehensive biomarker profiling to inform treatment selection.9,10 As Dr. Lin emphasized, this reality underscores the importance of delivering the most effective, biologically informed therapy upfront, when patients are most likely to derive meaningful benefit.2 In this context, CLDN18.2 represents a biologically defined and clinically relevant biomarker that may further refine patient selection for targeted and immunotherapy-based combination strategies.4,9
Conclusion
Emerging data from the phase 2 ILUSTRO trial highlight the potential of zolbetuximab-based triplet therapy as a biomarker-driven first-line strategy for patients with CLDN18.2-positive advanced G/GEJ cancer.3 In cohort 4B, zolbetuximab combined with mFOLFOX6 + nivolumab demonstrated encouraging PFS and response rates, particularly in patients with high CLDN18.2 expression and PD-L1 positivity, while maintaining a manageable safety profile.3 Although these findings remain exploratory, they underscore the importance of precision approaches in the frontline setting, where therapeutic impact may be greatest.1,2 Ongoing phase 3 evaluation in the LUCERNA trial will be critical to determining the role of zolbetuximab triplet therapy in the evolving treatment paradigm for advanced G/GEJ cancer.2,3

