CONFERENCE UPDATE: EADV 2024
Dupilumab treatment in younger children with AD may improve lifelong growth outcomes
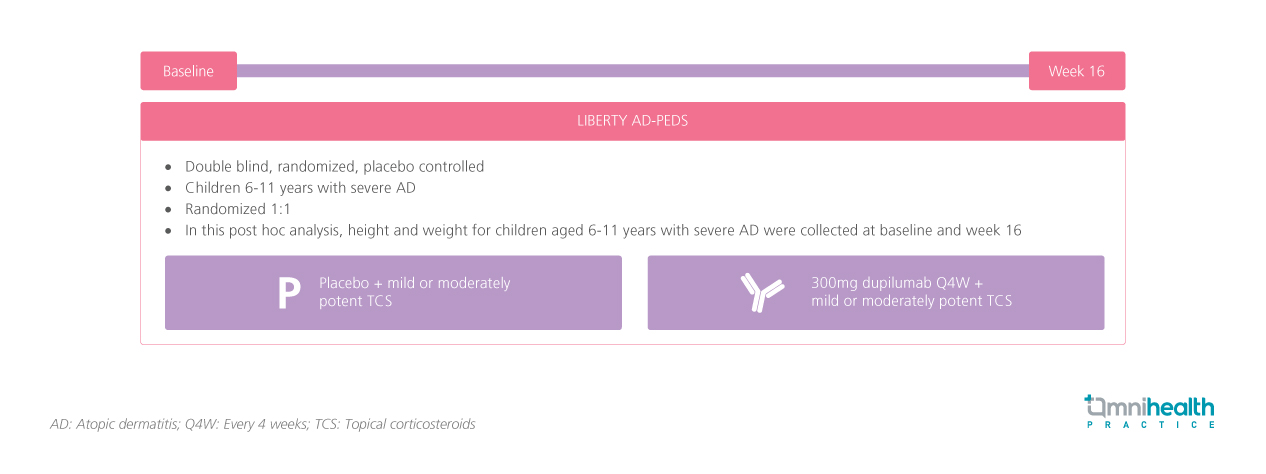
STUDY DESIGN
A 2007-2008 assessment of atopic dermatitis (AD) severity and height revealed that adolescents with moderate to severe AD had significantly higher odds of having a height <25th percentile on Center for Disease Control (CDC) growth reference chart.1 In particular, children with AD and in the ≤25th height percentile have a greater risk of having low bone mineral density and alkaline phosphatase (ALP) levels.1 The underlying mechanisms of this phenomenon remain unclear but may be linked to chronic sleep disruption associated with AD, the use of oral and topical glucocorticoids, and the effects of prolonged inflammation.1
Dupilumab was previously shown to significantly increase the levels of bone ALP in serum compared to placebo in children aged 6 to 11 with moderate-to-severe AD enrolled in a phase 3 trial and it’s open-label extension (OLE) study.1 In the LIBERTY-AD PEDS trial, the proportion of children aged 6 to 11 years with severe AD and lower stature who reached a ≥5-percentile improvement in height following 16 weeks of treatment with dupilumab compared to those in the placebo group was reported.1
LIBERTY-AD PEDS was a double-blind, randomized, placebo-controlled study that enrolled 304 children aged 6 to 11 years with severe AD.1 Participants were randomized 1:1 to receive 300mg dupilumab every 4 weeks or placebo, in addition to mild or moderately potent topical corticosteroids.1 The height and weight of the participants were collected at baseline and at week 16 in a post-hoc analysis.1
"Prompt and effective management of AD with dupilumab in younger children may have lifelong benefit in those who are below expected height by improving vertical growth"
Presented by Professor Alan Irvine Trinity College Dublin Dublin, Ireland
FINDINGS
| Baseline results: |
|
|
|
| Results at 16 weeks: |
|
|
|

