CASE REVIEW
Brexpiprazole augmentation achieving remission in MDD with anxiety features during long-term venlafaxine maintenance: A case report
Major depressive disorder (MDD) remains a challenging psychiatric condition, with nearly half of patients failing to achieve an adequate response to initial antidepressant therapy (ADT).1 Traditional augmentation strategies, including second-generation antipsychotics, may offer symptomatic relief but are often limited by adverse effects such as akathisia, sedation, and metabolic disturbances.2 In a recent interview with Omnihealth Practice, Dr. Miao, Yin-King May discussed a case of relapsed MDD with anxiety features in a patient who had been adherent to long-term venlafaxine therapy for over ten years. The condition was successfully managed through augmentation with low-dose brexpiprazole, a serotonin-dopamine activity modulator (SDAM), characterized by its ability to stabilize neurotransmitter systems while minimizing side effects.2,3 Its demonstrated efficacy, along with its tolerability across symptom domains of MDD such as anxiety and functional disturbances, underscores its clinical value.2,4
Background
The challenge of inadequate response in MDD
MDD is a complex and challenging condition to treat.1 Despite a wide array of available ADTs, a significant portion of patients do not achieve a satisfactory response.1 It is estimated that approximately 50% of individuals with MDD do not respond adequately to their initial ADT, and only about 35%-40% experience a full remission of symptoms within the first 8 weeks of treatment.1 A lack of response or a partial response can profoundly impact one's life, often resulting in decreased work productivity and a diminished quality of life (QoL).1 For patients with inadequate response to first-line medications, clinicians often turn to strategies like switching to a different ADT, or augmenting the current ADT with a non-ADT.2 The use of second-generation antipsychotics as adjunctive therapy has become a key strategy.2 However, their use can be limited by concerning side effects, such as akathisia, sedation, or weight gain.2 This has driven the search for newer agents with a more favorable balance of efficacy and tolerability.
Mechanism of action: A serotonin-dopamine activity modulator
Brexpiprazole is a therapeutic agent developed to meet this need as an adjunctive therapy for adults with MDD.3 Its therapeutic effects stem from its distinct pharmacological profile as a serotonin-dopamine activity modulator (SDAM).3 It has partial agonistic activity at dopaminergic D2 and serotonergic 5-HT1A receptors and is an antagonist at serotonergic 5-HT2A receptors.3 Unlike earlier antipsychotics, brexpiprazole fine-tunes and stabilizes key neurotransmitter systems involved in mood regulation through a combination of partial agonism and potent antagonism at several receptors.2,3
As a partial agonist at D2 and 5-HT1A receptors, brexpiprazole acts as a modulator.3 In brain regions where D2 activity is low, it provides a modest boost, but in regions where D2 activity is excessive, it acts as a brake, preventing overstimulation.3 Additionally, brexpiprazole serves as a potent antagonist at 5-HT2A receptors, which may contribute to its low incidence of neuromotor adverse effects.3 This unique combination of activities enables brexpiprazole to modulate multiple pathways simultaneously, contributing to a pharmacological profile that may offer improved tolerability compared with earlier adjunctive antipsychotics, such as aripiprazole.2,3 These properties position brexpiprazole as a well-supported option for augmentation in MDD. The following case shared by Dr. Miao illustrates the efficacy of brexpiprazole augmentation in sustaining recovery in a patient who experienced a relapse of depressive symptoms after more than 10 years of stability on venlafaxine monotherapy.
Case sharing
Ms. Z, a 61-year-old married woman with two sons, was diagnosed with MDD with anxiety features in June 2012. She had a longstanding pattern of being active in both her career and family life. She initially presented with anxiety, depressive affect, and somatic complaints during the perimenopausal period in 2012. At that time, she was undergoing hormone replacement therapy (HRT) and taking traditional Chinese medicine. She was started on venlafaxine 150mg daily, which resulted in significant improvement. Clinical stability was maintained even after cessation of HRT.
Throughout 2012 to 2020, she remained well-controlled on venlafaxine. She encountered marital stress and parenting challenges (elder son with mild autistic features, younger son with behavioral difficulties) as well as menopausal symptoms, but maintained stability with ongoing treatment. In 2020, she developed vasomotor symptoms (flushing, sweating) alongside psychosocial stressors, including bereavement (death of her father) and a family legal dispute over inheritance. Despite these stressors, she remained relapse-free until 2025, adapting over time through active coping, regular exercise, and social engagement.
In January of 2025, she relapsed with low mood, reduced motivation, and diminished drive despite adherence to venlafaxine. Brexpiprazole 0.5mg daily was initiated as augmentation to venlafaxine. At the one-month review in February of 2025, her condition had significantly improved. She reported restored mood, renewed enjoyment of daily activities, and full functional recovery. The regimen of venlafaxine extended-release 150mg daily and brexpiprazole 0.5mg daily has been maintained since.
Dr. Miao commented that Ms. Z’s decade-long stability on venlafaxine highlights the value of sustained pharmacotherapy in recurrent anxiety-depression. Her relapse despite adherence underscores that recurrence can still occur. Augmentation with brexpiprazole complements venlafaxine with its pharmacological profile as an SDAM, targeting both mood and anxiety symptoms, and rapidly restored symptom remission. Beyond symptomatic remission, Ms. Z regained occupational, social, and family functioning, reflecting the real-world clinical success of the intervention.
Discussion
Clinical efficacy: Robust improvement in depressive symptoms
The efficacy of adjunctive brexpiprazole in MDD demonstrated in the case has been established in clinical trials involving patients who had previously failed to respond to standard ADTs.
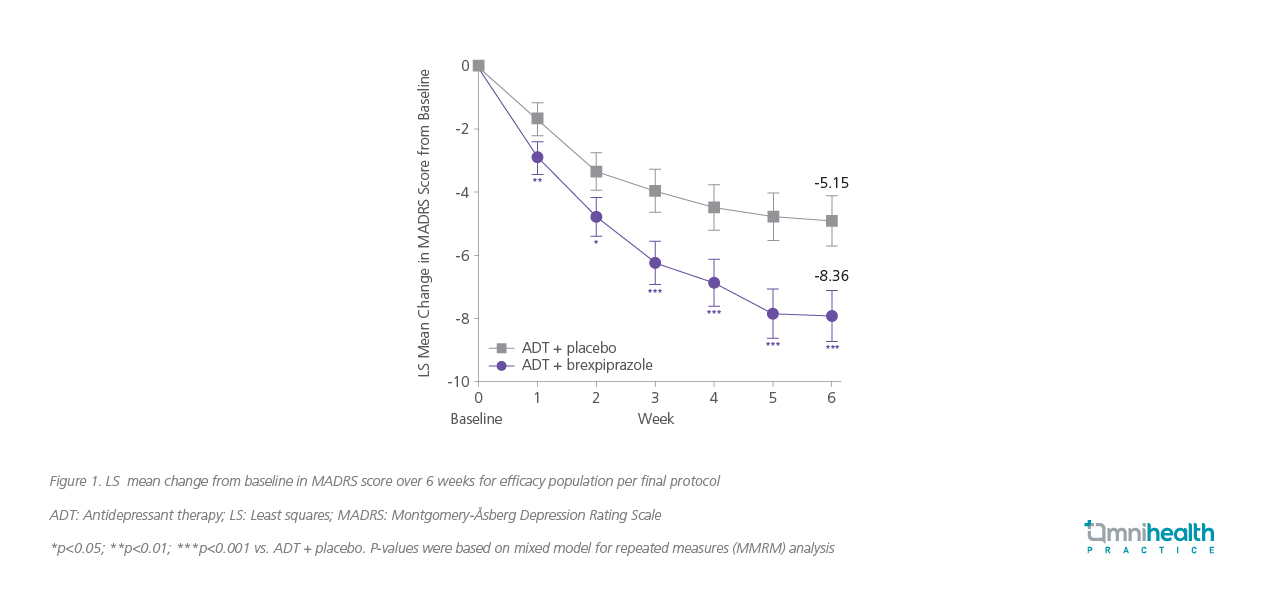
The Pyxis trial was a pivotal phase 3 study designed to assess the efficacy of adjunctive brexpiprazole.2 Patients with an inadequate response to 1-3 prior ADTs first entered an 8-week prospective phase during which they received a standard, open-label ADT.² Those who still did not respond adequately were then randomized to receive either adjunctive brexpiprazole 2mg or adjunctive placebo for 6 weeks.2 The primary objective was to evaluate the change in the Montgomery-Åsberg Depression Rating Scale (MADRS) total score from baseline to week 6.2 Patients receiving adjunctive brexpiprazole experienced a statistically significant and clinically meaningful greater reduction in MADRS total score at week 6 compared with placebo (least squares [LS] mean change of -8.36 vs. -5.15; p=0.0002) (figure 1).2 Separation from placebo was apparent as early as the first week of treatment and was maintained throughout the study.2
Targeting symptom clusters: Anxiety and functional domains
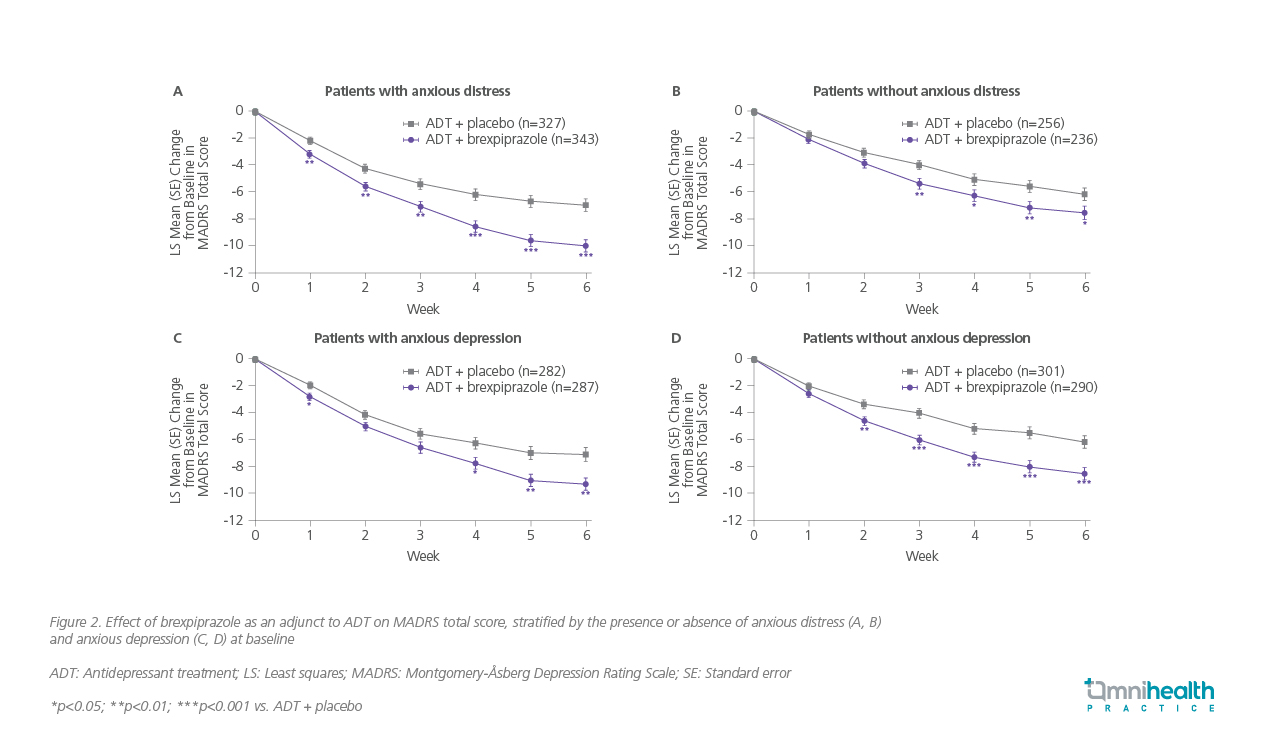
Beyond overall efficacy demonstrated in the case, clinical data provide insights into brexpiprazole’s effects on specific symptom clusters within MDD. Anxiety frequently coexists with MDD.5 In the Pyxis trial, adjunctive brexpiprazole demonstrated a statistically significant improvement in anxiety symptoms compared to placebo, as measured by the Hamilton Anxiety Rating Scale (HARS) total score, with a LS mean change of -3.79 vs. -2.70 for placebo (p=0.0376).2 Dr. Miao remarked that “Treatment must extend beyond symptomatic remission to resolve key drivers like anxious distress to achieve functional recovery in MDD.“ In a pooled analysis of phase 3 studies, adjunctive brexpiprazole combined with ADT demonstrated significant improvement vs. ADT + placebo as early as week 1 in patients with anxious distress, with benefits maintained throughout the 6-week treatment period (LS mean difference at week 6: -3.00; 95% CI: -4.29 to -1.71; p<0.0001). Improvements were also seen in patients without anxious distress, albeit from week 3 onward (LS mean difference at week 6: -1.38; 95% CI: -2.71 to -0.05; p=0.043).6 Similar benefits were observed when stratified by the presence or absence of anxious depression (figure 2).6 Furthermore, brexpiprazole also demonstrated a significant improvement in patient functioning, the key secondary endpoint measured by the Sheehan Disability Scale (SDS) mean score, with a LS mean change of -1.35 vs. -0.89 for placebo (p=0.0349).² This indicates that the reductions in depressive symptoms translated into tangible improvements in patients' social and family lives.
In a post-hoc, pooled analysis of four short-term, randomized, double-blind, placebo-controlled trials in adults with MDD and inadequate response to 2-4 ADTs, adjunctive brexpiprazole 2mg daily (n=486) added to ADT demonstrated significantly greater improvement than placebo + ADT (n=585) across multiple symptom clusters of depression over 6 weeks—including core symptoms (effect size [ES]: 0.36), anhedonia (ES: 0.43), dysphoria (ES: 0.27), retardation (ES: 0.32), vegetative symptoms (ES: 0.29), loss of interest (ES: 0.30), and lassitude (ES: 0.33)(all p<0.0001).4 Improvements were observed from week 1 onwards and were maintained through week 6.4 These data indicate that adjunctive brexpiprazole 2mg daily may benefit a range of symptom clusters in MDD with inadequate response to ADTs.
Safety and tolerability: A favorable profile
Across the clinical trial program, adjunctive brexpiprazole has demonstrated a favorable and manageable safety profile.1,2 In Pyxis trial, weight gain was reported in 8.0% of patients on brexpiprazole vs. 3.1% on placebo, with an average gain of 1.64kg over 6 weeks in the brexpiprazole group.2 Akathisia occurred in 7.4% of patients vs. 1.0% for placebo.² Sedation-related events were minimal, with somnolence reported in just 4.3% of patients and fatigue in 1.6%, rates comparable to placebo.² Similarly, other activating side effects like restlessness (3.2%) and insomnia (2.1%) were uncommon.2 This profile aligns with its mechanism of action, which combines lower intrinsic activity at the D2 receptor with moderate affinity for the histamine H1 receptor.³ This balance of robust efficacy and manageable side effects supports its role as an evidence-based augmentation strategy for patients with MDD who have an inadequate response to standard ADT. The 2023 Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines further reinforce this recommendation, listing brexpiprazole as a first-line augmentation option for MDD, supported by Level 1 evidence for both efficacy and safety.7
Conclusion
Adjunctive brexpiprazole represents a well-supported therapeutic option for patients with MDD who have inadequate response to standard ADTs. Its distinct SDAM mechanism allows for nuanced modulation of dopaminergic and serotonergic pathways, resulting in significant improvements in depressive symptoms.1-3 The case sharing from Dr. Miao highlights the efficacy of brexpiprazole augmentation in a patient with long-term venlafaxine maintenance who experienced a relapse of depressive symptoms. Such an augmentation strategy may be particularly valuable for late-life patients with recurrent or treatment-resistant depressive episodes.

