CONFERENCE UPDATE: AASLD 2025
Sustained improvement in pruritus among patients with PBC treated with seladelpar: Results from the 30-month ASSURE Study
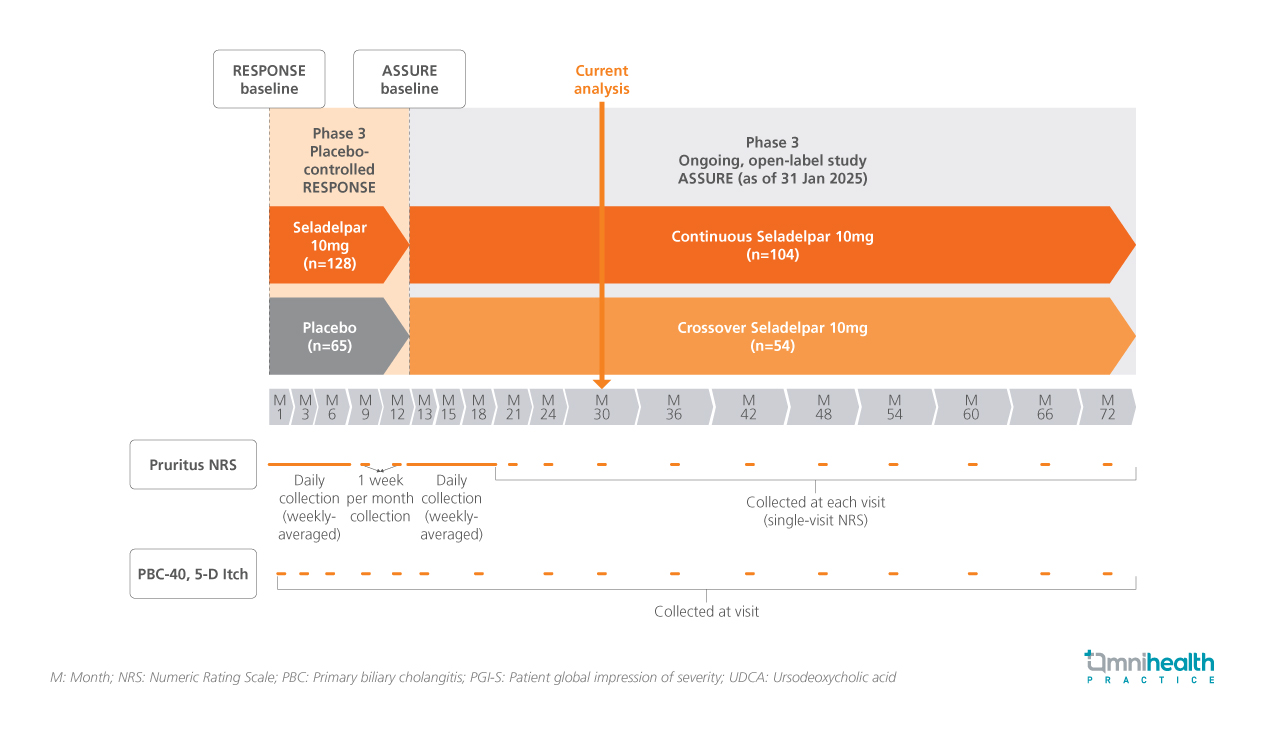
STUDY DESIGN
Primary biliary cholangitis (PBC) is a chronic, progressive, autoimmune cholestatic liver condition that mainly affects women over the age of 40.1 Many patients—up to 80%—experience cholestatic pruritus, a symptom that can severely diminish quality of life and, in extreme cases, prompt consideration of liver transplantation even when liver function is still preserved.1 Conventional treatments such as bile acid sequestrants, antihistamines, and off-label rifampin often provide incomplete or inconsistent symptom relief, highlighting the need for better therapeutic options.1 Seladelpar, a first-in-class selective PPARδ agonist, previously demonstrated notable improvements in pruritus and biochemical markers in the pivotal RESPONSE study.1
During the presentation, long-term pruritus outcomes from the phase 3 RESPONSE study of seladelpar in adults with PBC and interim results from its ongoing open-label ASSURE studywere shown.1 In the RESPONSE study, pruritus scores using the numeric rating scale (NRS) were gathered daily for the first six months, followed by weekly collections for one week each month over a span of twelve months.1 After transitioning to the ASSURE phase, NRS was recorded daily for another six months and during scheduled clinic appointments thereafter.1 Additional assessments included the PBC-40 and 5-D Itch questionnaires.1 As of January 312025, patients exhibiting moderate to severe pruritus (NRS ≥4) at baseline were evaluated based on their randomization to either seladelpar 10mg or placebo in the RESPONSE phase and were subsequently categorized as continuous or crossover seladelpar 10mg groups in ASSURE.1 Baseline characteristics were well-balanced between the seladelpar (n=49) and placebo (n=23) arms in patients with moderate to severe pruritus.1
Outcomes assessed included changes in pruritus measured by the NRS, the 5-D Itch scale, and the PBC-40 itch domain over 30 months of treatment.1 The percentage of patients achieving a clinically meaningful improvement of ≥3 points on the NRS was also evaluated, along with comparisons between single-visit NRS scores and weekly averages in the ASSURE phase.1
FINDINGS
|
Change in pruritus: |
|
|
Clinically meaningful improvement in the NRS (≥3-point change): |
|
|
Comparability of single-visit NRS and weekly averaged NRS in ASSURE: |
|
“Seladelpar led to sustained, clinically meaningful improvement in pruritus in ASSURE with up to 30 months of treatment”Dr. Gideon Hirschfield
The Autoimmune and Rare Liver Disease Programme,
Toronto General Hospital, Toronto, Canada

