EXPERT INSIGHT
Lighting the path: How sentinel nodes guide EC care
Endometrial cancer (EC), the most common gynecologic malignancy and the sixth most frequently diagnosed cancer among women worldwide, is increasingly prevalent in Asia, which accounted for nearly 40% of new cases in 2020.1 Effective management of early-stage disease continues to center on surgery, typically hysterectomy with bilateral salpingo-oophorectomy, where accurate lymph node assessment remains critical for prognosis and guiding adjuvant therapy.2 Over the past decade, sentinel lymph node (SLN) mapping using indocyanine green (ICG) and near-infrared (NIR) fluorescence imaging has emerged as a transformative alternative to lymphadenectomy, offering precise nodal assessment with markedly reduced surgical burden.3,4 In a recent expert discussion with Omnihealth Practice, Professor Nirmala Kampan, a gynecologic oncologist and pioneer of SLN mapping in Malaysia, shared her perspectives on the evolving role of this technique, from evidence-based outcomes to future innovation and directions in patient-centered care in EC.
EC on the rise: Addressing unmet needs
EC affects approximately 3% of women over their lifetime, with a median age at diagnosis of 61.5 Alarmingly, its incidence has surged by 132% in the past three decades, with cases among women under 40 having doubled.5 This upward trend is closely tied to population aging and modifiable risk factors such as obesity and sedentary lifestyles.5 With most patients diagnosed at an early stage, there is a growing need for precise, yet less invasive, strategies to evaluate lymph node involvement.5
Accurate assessment of lymph node status is central to staging and prognosis in EC.2 In clinically apparent early-stage disease, the traditional standard of care has been surgical staging with hysterectomy, bilateral salpingo-oophorectomy, and evaluation of regional lymph nodes.4 Yet, the optimal extent of lymphadenectomy—pelvic alone vs. combined pelvic and para-aortic, selective sampling vs. complete dissection—has long been debated in gyne-oncology.4 While full lymphadenectomy provides accurate staging, it also carries considerable morbidity, including longer operative times, risk of nerve or vessel injury, and long-term complications such as lymphocysts and lymphedema.3,6 According to Prof. Nirmala, these challenges have driven the adoption of SLN mapping as a less morbid yet reliable alternative for nodal assessment in early-stage disease.
Sentinel node mapping: Precision without added burden
SLN mapping with ICG, combined with minimally invasive hysterectomy, is rapidly gaining ground as the preferred approach for early-stage EC.4 By targeting the first node(s) receiving lymphatic drainage from the primary tumor, SLN mapping provides a reliable marker of regional metastatic spread while limiting the morbidity associated with full lymphadenectomy.2
A variety of tracers have been explored for SLN mapping.6 Blue dyes, such as isosulfan blue and methylene blue are approved by the Food and Drug Administration (FDA), but they are limited by inconsistent availability, potential allergic reactions, and interference with oxygen monitoring.3 Radiolabeled isotopes offer deeper tissue penetration, but require preoperative lymphoscintigraphy or three-dimensional single photon emission computed tomography with integrated computed tomography (SPECT/CT), expose patients to radiation, and add logistical complexity through coordination with nuclear medicine.3 By contrast, ICG visualized with NIR fluorescence imaging has emerged as the favored tracer, combining high detection rates with ease of use and an excellent safety profile, particularly in patients with obesity.3,4
The technical approach has also evolved to maximize SLN detection.3 ICG, a water-soluble tricarbocyanine dye, fluoresces in the NIR spectrum.3 When diluted to 0.5mg/mL-1.25mg/mL and injected in volumes of 2mL-4mL, it provides clear visualization of lymphatic pathways.3 NIR imaging systems, adaptable to open, laparoscopic, and robotic surgery, detect the dye’s 830nm emission and provide real-time intraoperative guidance (figure 1).3
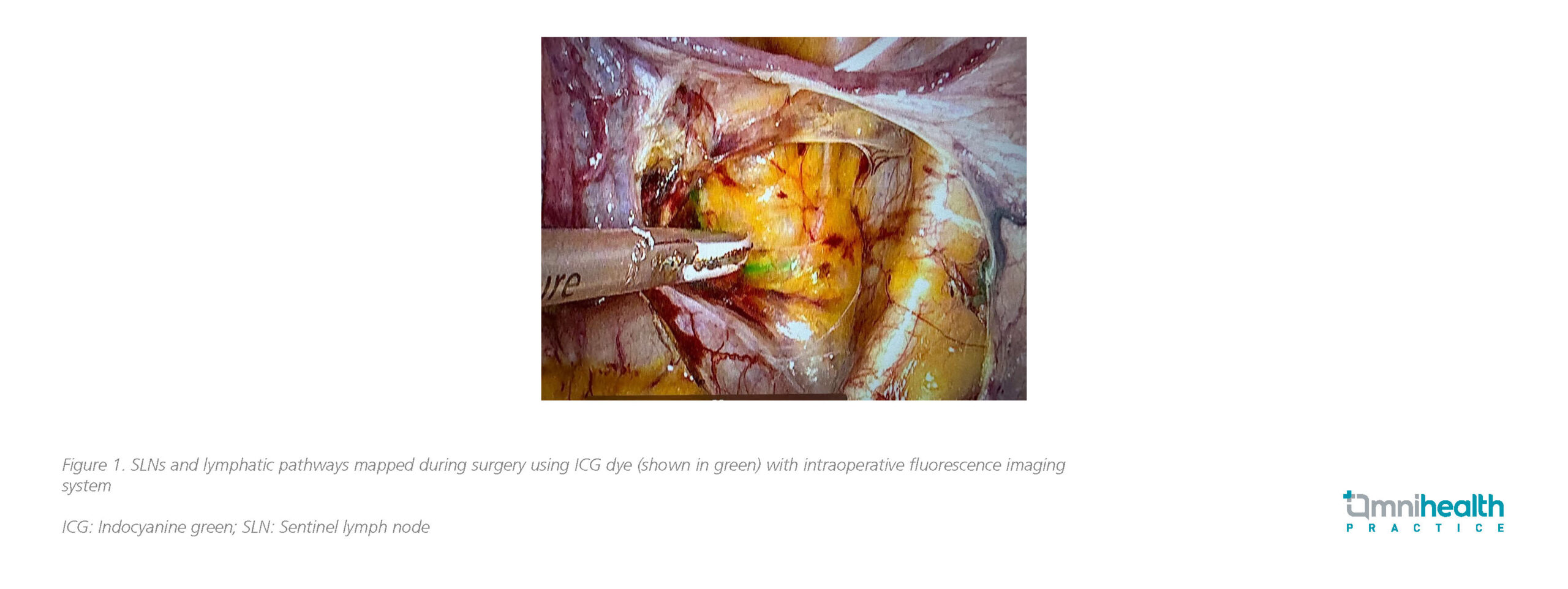
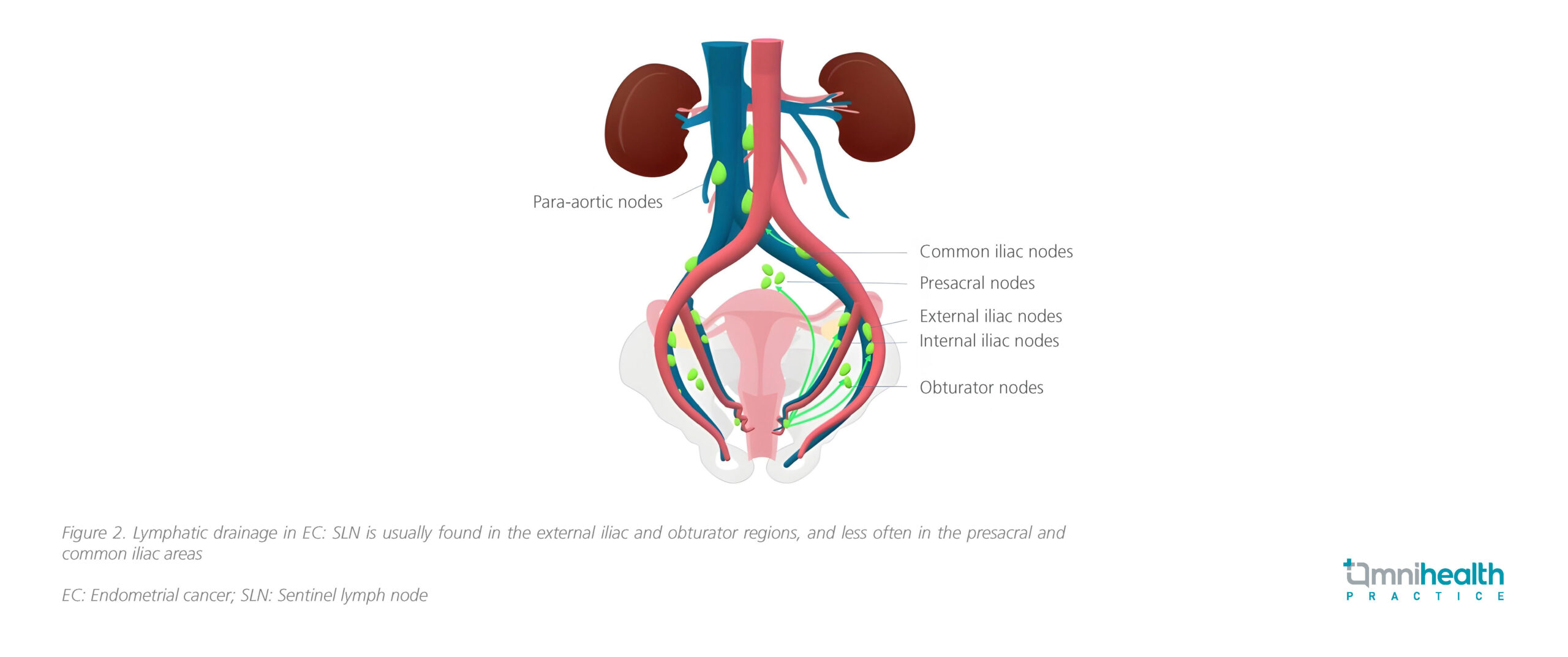
Cervical injection has become the most widely adopted method due to its simplicity and consistently high detection rates.3 Slow submucosal or superficial stromal injection further improves tracer uptake into the lymphatic pathway leading to the SLN (figure 2) and reduces background staining of pelvic tissues.3
Despite its advantages, SLN mapping is not always successful.5 Failures, occurring in up to 6% of cases, are typically due to bulky tumors obstructing lymphatic drainage or patient-related factors such as obesity.5 In these situations, international guidelines recommend side-specific lymphadenectomy, particularly in patients with intermediate- or high-risk disease.5
To optimize SLN mapping and reduce false negatives, the National Comprehensive Cancer Network (NCCN) Guidelines 2025 outline several key principles:3
- SLN mapping is recommended for surgical staging in patients with apparent uterine-confined EC, where no metastasis is detected on imaging or at exploration
- Superficial and deep cervical dye injection is validated as a reliable mapping method, with ICG now considered the preferred tracer
- The success of SLN mapping depends on surgeons’ expertise and meticulous attention to technical detail
- When combined with ultrastaging, SLN mapping improves detection of nodal metastases while maintaining low false-negative rates compared with full lymphadenectomy
- Successful mapping requires strict adherence to the SLN algorithm, which requires a side-specific dissection in cases of failed mapping and removal of any suspicious or enlarged nodes
- If mapping fails in one hemi-pelvis, a side-specific lymphadenectomy is recommended; however, in centers with expert pathology support, frozen section may be used to assess myometrial or cervical invasion, potentially sparing lymphadenectomy when myoinvasion is absent
- SLN identification should be performed before hysterectomy, except in cases where a bulky uterus must be removed to gain access to the pelvic vessels and nodes
- Serial sectioning and immunohistochemistry (IHC) enhance the detection of low-volume metastasis (including micrometastases and isolated tumor cells)
Mapping success: Clinical evidence and real-world outcomes
Clinical data strongly support the role of SLN mapping with ICG in EC.8 Meta-analyses of more than 2,000 patients demonstrate an overall detection rate of 95.6% and bilateral detection of 76.5% with a negative predictive value approaching 100% in grade 1-2 tumors and remaining above 99% even in grade 3 disease.8 Further analysis of 5,820 patients showed no significant differences in overall survival, progression-free survival, or recurrence rates, compared with full lymphadenectomy, while offering perioperative advantages such as reduced blood loss and operative time.9
Real-world experience mirrors these findings. At her center, Prof. Nirmala reported that women undergoing minimally invasive surgery with SLN mapping are typically discharged on the second or third postoperative day, significantly improving quality of life compared with longer stays associated with open surgery and full lymphadenectomy. Reflecting on her team's performance, “Our team at HCTM-UKM has achieved SLN detection rates exceeding 80%, consistent with international benchmarks.” She noted that the procedure adds only a few minutes to a laparoscopic hysterectomy, and when combined with frozen section analysis, it allows real-time intraoperative decision making, an approach that enhances precision, safety, and recovery. Together, these clinical and real-world outcomes indicate that SLN mapping with ICG is both safe and patient-friendly, supporting its adoption as a standard approach in early-stage EC.
Shaping the future of EC care
The next frontier for EC care lies in personalized oncology.3 Prof. Nirmala noted that molecular biomarkers such as POLE, MMR, MSI, and PD-1/PD-L1 are being investigated to refine risk stratification and guide surgical planning. This approach could allow women with low-risk disease to avoid extensive procedures while ensuring that high-risk groups receive appropriately aggressive treatment.
Advances in surgical technology are complementing these molecular insights.10,11 “Advances in surgical technology are broadening the scope of minimally invasive care,” Prof. Nirmala remarked, underscoring how new platforms and techniques are reshaping possibilities in gynecologic oncology. Robotic-assisted surgery shortens the learning curve for minimally invasive procedures, provides enhanced ergonomics, and enables safer, more controlled dissection in patients with complex anatomy or significant comorbidities.10 Novel approaches, such as vaginal natural orifice transluminal endoscopic surgery (vNOTES) are being tested for scarless procedures.11 Taken together, these innovations point toward a future in gyne-oncology that is increasingly precise, safe, and accessible.10,11
At the same time, prevention and early detection remain indispensable.5 EC often produces recognizable warning signs, most notably abnormal uterine bleeding.5 Whether unusually heavy menses, intermenstrual bleeding, or postmenopausal bleeding, such symptoms warrant prompt investigation.5 The diagnostic pathway is relatively straightforward, beginning with a transvaginal ultrasound to assess endometrial thickness, followed by histological examination.5 Outpatient hysteroscopy allows direct sampling of suspicious lesions and is recommended when ultrasound suggests focal endometrial pathology or in women with recurrent symptoms.5 Particular vigilance is needed in high-risk groups, including women with polycystic ovarian syndrome, chronic anovulation, obesity-related metabolic disorders, or those taking tamoxifen for breast cancer.5
Public awareness remains a critical barrier.5 Prof. Nirmala emphasized that many women attribute menstrual irregularities to stress or benign causes, delaying medical care until the disease is more advanced. She noted that EC is increasingly being diagnosed in women in their 30s, where early recognition not only enables timely treatment but also offers the possibility of fertility-preserving options in selected early-stage cases. Prompt diagnosis often permits curative treatment while sparing patients the complications and more aggressive therapies associated with advanced disease.
Lastly, risk factor modification remains crucial, with obesity identified as the most significant modifiable risk factor.5 Successful management of obesity, through regular physical activity, a healthy diet, or pharmacotherapy, is essential to mitigate this risk.5 Additional lifestyle factors, such as limiting alcohol intake and controlling comorbid metabolic disorders, may further reduce disease burden.12,13 By addressing these modifiable risks, clinicians and patients can play an active role in preventing EC and complement the benefits of early detection and personalized treatment.5,13
Conclusion
In conclusion, for women with early-stage EC, advances in surgical care are opening new possibilities.2,3 SLN mapping represents a new paradigm, combining oncologic precision with reduced morbidity and faster recovery.2,3 Prof. Nirmala emphasized that broader adoption would depend on standardized protocols, structured surgeon training, and solutions to improve access to both technology and expertise. Equally important, prevention and public awareness remain critical, as early detection at the earliest possible stage is key to improving cure rates, minimizing treatment-related morbidity, and reducing mortality.5

