CONFERENCE UPDATES: ESC 2025
Inclisiran-based strategy improves LDL-C goal attainment and safety: Results from the phase 4 VICTORION-Difference trial
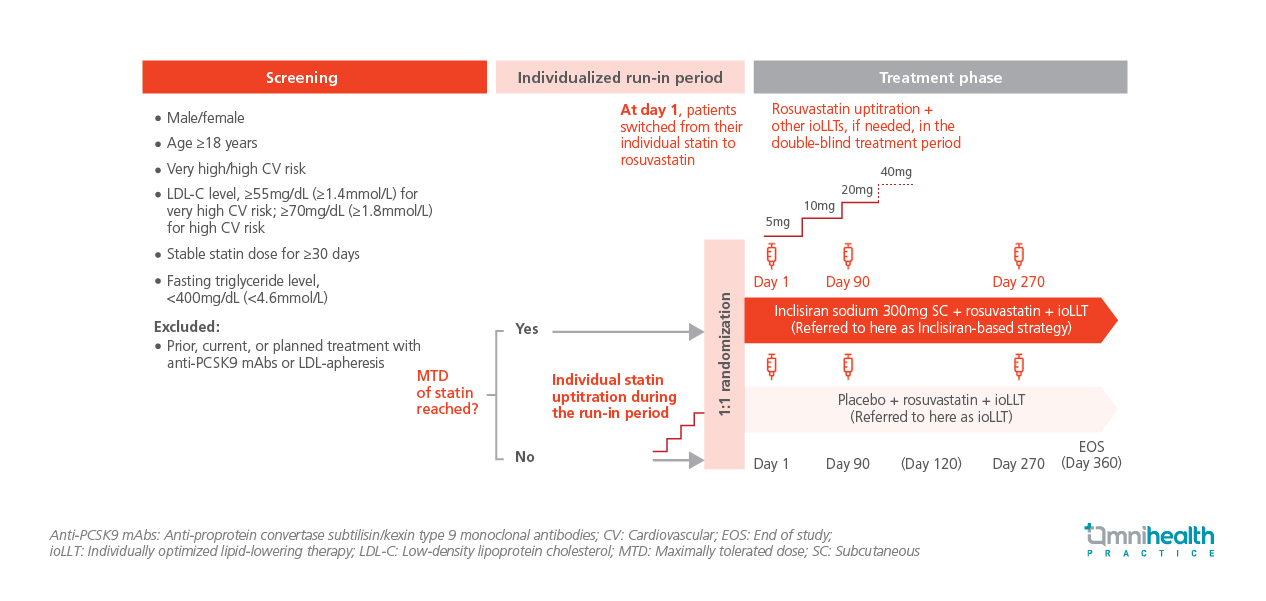
STUDY DESIGN
Atherosclerotic cardiovascular disease (ASCVD) remains a substantial health burden, with low-density lipoprotein cholesterol (LDL-C) established as a causal risk factor in both its development and progression.1 Despite maximally tolerated dose (MTD) of statin therapy and further therapy escalation, many patients do not achieve recommended LDL-C targets, often due to tolerability concerns.1
Inclisiran is a small interfering ribonucleic acid (siRNA) targeting hepatic proprotein convertase subtilisin/kexin type 9 messenger ribonucleic acid (PCSK9 mRNA) that is administered twice yearly and offers sustained and effective LDL-C reductions.1 The VICTORION-Difference trial assessed the efficacy, safety, tolerability, and quality-of-life (QoL) outcomes of an inclisiran-based strategy vs. individually optimized lipid-lowering therapy (ioLLT) in patients with high or very high cardiovascular (CV) risk who fail to achieve guideline-recommended LDL-C targets despite statin MTD.1
This phase 4, double-blind, placebo-controlled study enrolled a total of 1,770 patients across 133 sites in 8 European countries.1 Prior to randomization, patients underwent a statin run-in period during which they were switched to rosuvastatin and up-titrated to MTD if guideline-recommended LDL-C levels were not attained.1 Eligible patients were ≥18 years old with high or very high CV risk, with fasting triglyceride level <400mg/dL and on a stable statin dose for ≥30 days.1 At randomization, participants were assigned 1:1 to either inclisiran sodium 300mg subcutaneous (SC) injection + rosuvastatin + ioLLT (inclisiran-based strategy) or placebo + rosuvastatin + ioLLT (ioLLT strategy).1 Baseline demographics and clinical characteristics were well-balanced between arms.1 Patients were followed up for 360 days with assessments at multiple timepoints.1
The primary endpoint was the proportion of patients achieving LDL-C goals at day 90.1 Key secondary endpoints included the percentage change in mean LDL-C from baseline to day 360 and the proportion of patients with muscle-related adverse events (AEs) over the same period.1 Additional endpoints assessed patient-reported outcomes (PROs), including changes in pain severity, interference scores, and the annualized number of days during which patients experienced pain.1
FINDINGS
| Primary endpoint : |
|
|
|
| Key efficacy endpoint : |
|
|
| Key safety endpoint : |
|
|
|
| Patient-reported outcomes : |
|
|
|

