CONFERENCE UPDATES: ESC 2025
Baxdrostat reduces SBP in uncontrolled and resistant hypertension: Results from the phase 3 BaxHTN trial
STUDY DESIGN
Individuals with uncontrolled or resistant hypertension face a significant risk of cardiovascular complications, mortality, and negative renal outcomes.1 Aldosterone dysregulation plays a crucial role in the development of uncontrolled and resistant hypertension, as well as in the organ damage associated with hypertension.1 Although mineralocorticoid receptor antagonists (MRAs) can attenuate aldosterone-mediated damage, their clinical application is limited by dose-dependent adverse effects.1 Baxdrostat is a highly selective and potent aldosterone synthase inhibitor.1 Its efficacy and safety in patients with uncontrolled or resistant hypertension were assessed in the phase 3 BaxHTN trial.1
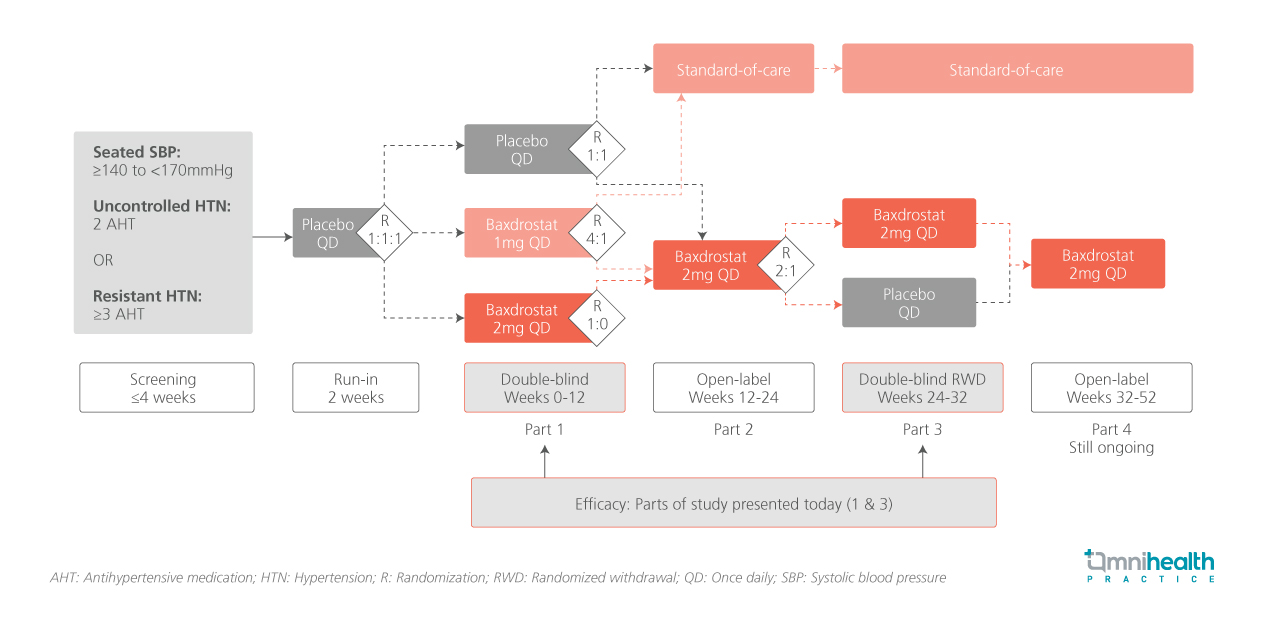
The BaxHTN trial is a double-blind, randomized, placebo-controlled study incorporating a 12-week double-blind phase, followed by an open-label period and a randomized withdrawal (RWD) phase.1 The trial enrolled 794 adult patients with uncontrolled (27%) or resistant (73%) hypertension.1 The mean age of participants was 61 years, and 62% were men.1 Key inclusion criteria were mean seated systolic blood pressure (SBP) of 140mmHg-169mmHg despite stable therapy with ≥2 antihypertensive agents (uncontrolled hypertension) or ≥3 agents (resistant hypertension), including a diuretic, for ≥4 weeks before screening, estimated glomerular filtration rate (eGFR) ≥45mL/min/1.73m², and serum potassium ≥3.5mmol/L and <5.0mmol/L.1 Patients were excluded if they were using MRAs or potassium-sparing diuretics, had secondary hypertension, uncontrolled diabetes, cardiovascular or cerebrovascular events within 6 months, or persistent atrial fibrillation.1
Participants were randomized 1:1:1 to receive baxdrostat 1mg once daily (n=264), baxdrostat 2mg once daily (n=266), or placebo (n=264), in addition to their background antihypertensive therapy, for 12 weeks.1 This initial phase (part 1) was followed by a 12-week open-label phase (part 2) designed to collect safety data: patients already on 2mg continued treatment, while those previously on 1mg or placebo were re-randomized to either 2mg or standard-of-care therapy, providing a comparator for longer-term safety.1 In part 3, an 8-week double-blind withdrawal phase (weeks 24-32), patients receiving 2mg baxdrostat were re-randomized to continue treatment or switch to placebo.1 Part 4, which is still ongoing, is a 20-week open-label phase collecting additional safety data.1
The primary endpoint was the change in seated office SBP from baseline to week 12 (during the double-blind, placebo-controlled period).1 The key secondary endpoint was the change in SBP during the randomized withdrawal phase (weeks 24-32), whilst the key exploratory endpoint was the mean 24-hour and night-time ambulatory SBP.1
FINDINGS
|
Primary endpoint: |
|
|
Key secondary endpoint: |
|
|
Key exploratory endpoint: |
|
|
Safety: |
|

