CONFERENCE UPDATE: ASH 2024
Acalabrutinib + BR delivers significant PFS benefit in untreated older patients with MCL: Updated analysis from the ECHO trial
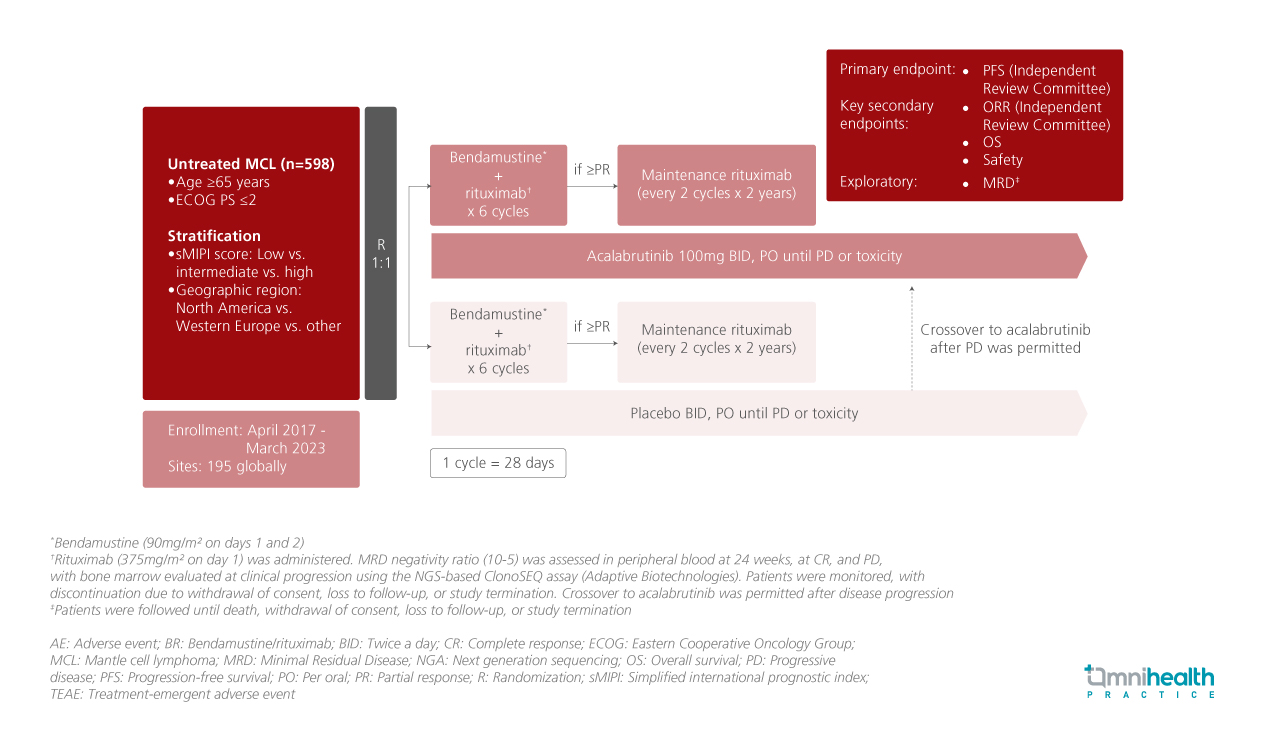
STUDY DESIGN
Older or unfit patients with mantle cell lymphoma (MCL) often do not tolerate intensive first-line therapies due to poor tolerability.1 In such cases, bendamustine-rituximab (BR) combination is the most commonly used first-line regimen.1 While adding Bruton's tyrosine kinase inhibitors (BTKi), such as ibrutinib, to BR has demonstrated improved progression-free survival (PFS), this approach has not translated into an overall survival (OS) benefit, primarily due to toxicity.1
Acalabrutinib, a next-generation BTKi with fewer off-target effects, was introduced to address this limitation.1 The ECHO trial evaluated the efficacy and safety of acalabrutinib + BR (ABR) in older patients with previously untreated MCL.1 In the primary analysis, ABR was shown to significantly improve PFS compared to placebo + BR (PBR) in older patients with previously untreated MCL, with a trend favoring acalabrutinib in OS.1
The ECHO trial was a phase 3, randomized, multicenter, double-blind, placebo-controlled study enrolling patients aged 65 years or older with previously untreated MCL.1 Patients with an Eastern Cooperative Oncology Group (ECOG) performance status of 0-2 were included and stratified by the simplified international prognostic index (sMIPI) score and geographic regions.1 Participants were randomized 1:1 to receive either ABR (n=299) or PBR (n=299).1 The median age across both arms was 71 years; most patients were male.1 The updated analysis presented additional efficacy (PFS among subgroups and MRD data) and safety results (including exposure-adjusted incidence rates and timing of adverse events [AEs]).1
"The addition of acalabrutinib to BR significantly reduced the risk of disease progression or death in older patients with previously untreated MCL, including those with high-risk features
Dr. Martin Dreyling
Medizinische Klinik III,
Klinikum der Universitaet München,
Munich, Germany
FINDINGS
| Efficacy endpoints: |
| • The additional efficacy endpoints presented included PFS among subgroups and MRD data1 |
| • PFS benefit was consistent across subgroups, including those with high-risk disease characteristics, such as TP53 mutation (HR=0.88; 95% CI: 0.42-1.78), high Ki-67 index (HR=0.63; 95% CI: 0.45-0.89), and blastoid/pleomorphic histology (HR=0.59; 95% CI: 0.33-1.07)1 |
| • Among evaluable patients who were MRD-negative at the end of induction (24 weeks), the rate of conversion to MRD-positive status during maintenance was lower in ABR arm (6%; n=11/188) compared to the PBR arm (15%; n=26/117)1 |
| • Conversely, among evaluable patients who were MRD-positive at the end of induction, 37.5% (n=3/8) in the ABR arm and 20% (n=3/15) in the PBR arm converted to MRD-negative status during maintenance1 |
| Safety: |
| • The incidence of AEs was similar between arms during induction but higher in the ABR arm during the maintenance and monotherapy phase1 |
| • When adjusted for exposure, the incidence rates per 100 person-years reduced the differences between ABR and PBR for clinically relevant TEAEs: 32.6 vs. 29.3 for grade ≥3 serious TEAEs, 4.2 vs. 4.0 for grade 5 TEAEs, and 15.5 vs. 12.4 for TEAEs leading to discontinuation. These differences may reflect the longer median exposure to acalabrutinib (28.6 vs. 24.6 months)1 |

