CONFERENCE UPDATE: ESMO 2024
Enzalutamide + Ra223 shows rPFS benefits in asymptomatic or mildly symptomatic patients with bone-metastatic mCRPC: Results from the PEACE-3 trial
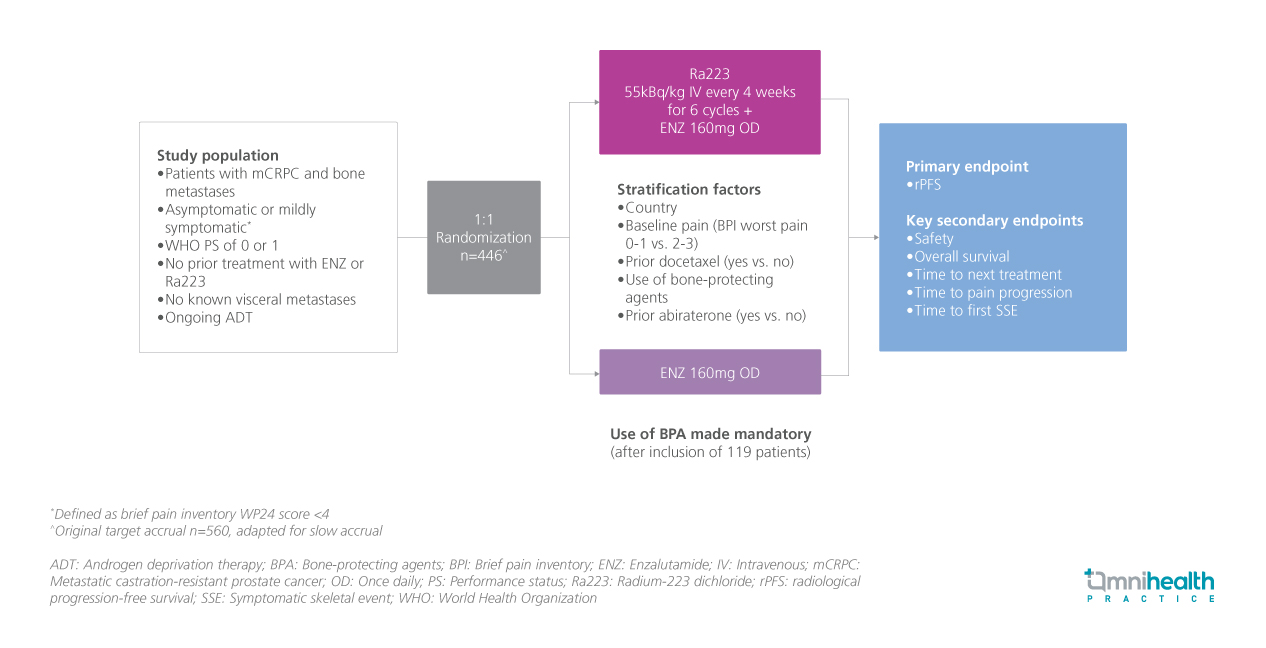
STUDY DESIGN
Abiraterone and enzalutamide (ENZ) are established first-line treatments for patients with metastatic castration-resistant prostate cancer (mCRPC) who progress on androgen deprivation therapy (ADT).1 Radium-223 dichloride (Ra223), an alpha particle-emitting calcium mimetic, selectively targets bone metastases by inducing double-strand deoxyribonucleic acid (DNA) breaks.1 The ERA-223 trial, which tested the combination of abiraterone with Ra223, found no improvement in symptomatic skeletal event-free survival or overall survival (OS).1 No combination so far has been proven in both radiological progression-free survival (rPFS) and OS as a first-line mCRPC treatment.1 In response, the PEACE-3 trial, a collaboration of EORTC, CTI, CUOG, LACOG, and UNICANCER, was launched to assess whether combining ENZ with Ra223 (ENZ + Ra223) could enhance cancer progression outcomes compared to ENZ alone in mCRPC patients.1
The PEACE-3 trial is a randomized, multicenter, open-label, phase 3 trial that recruited men with mCRPC and bone metastases who were receiving ongoing ADT, with no known visceral metastasis and no prior treatment with either ENZ or Ra223 (n=446).1 Participants were randomized 1:1 to receive either ENZ 160mg once daily (n=224) or a combination of ENZ 160mg once daily with intravenous Ra223 every 4 weeks for 6 cycles (ENZ + Ra223) (n=222).1
The primary endpoint was rPFS.1 Key secondary endpoints included safety, OS, time to subsequent systemic therapy, time to pain progression, and time to first symptomatic skeletal event (SSE).1 This study reports the first results of PEACE-3 trial after a median follow-up of 42.2 months.1
FINDINGS
| Primary endpoints: |
|
|
|
| Secondary endpoints: |
|
|
|
|
|
| Safety: |
|
|
|
|
|
*The pre-set level of significance for interim analysis was ≤0.0034
”These results support the combination of ENZ + Ra223 (+ a bone-protecting agent) as a potential new first-line mCRPC treatment option for patients with prostate ancer and bone metastases who have not received a prior androgen receptor-pathway inhibitor"
Professor Silke Gillessen
Oncology Institute of Southern Switzerland,
Bellinzona, Switzerland

